Bacterial GCN5-Related N-Acetyltransferases: From Resistance to Regulation
- PMID: 26818562
- PMCID: PMC4795176
- DOI: 10.1021/acs.biochem.5b01269
Bacterial GCN5-Related N-Acetyltransferases: From Resistance to Regulation
Abstract
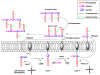
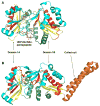
The GCN5-related N-acetyltransferases family (GNAT) is an important family of proteins that includes more than 100000 members among eukaryotes and prokaryotes. Acetylation appears as a major regulatory post-translational modification and is as widespread as phosphorylation. N-Acetyltransferases transfer an acetyl group from acetyl-CoA to a large array of substrates, from small molecules such as aminoglycoside antibiotics to macromolecules. Acetylation of proteins can occur at two different positions, either at the amino-terminal end (αN-acetylation) or at the ε-amino group (εN-acetylation) of an internal lysine residue. GNAT members have been classified into different groups on the basis of their substrate specificity, and in spite of a very low primary sequence identity, GNAT proteins display a common and conserved fold. This Current Topic reviews the different classes of bacterial GNAT proteins, their functions, their structural characteristics, and their mechanism of action.
Conflict of interest statement
The authors declare no competing financial interest.
Figures
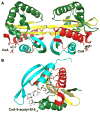
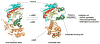
Similar articles
-
A bacterial acetyltransferase capable of regioselective N-acetylation of antibiotics and histones.Chem Biol. 2004 Apr;11(4):565-73. doi: 10.1016/j.chembiol.2004.03.017. Chem Biol. 2004. PMID: 15123251
-
Small-Molecule Acetylation by GCN5-Related N-Acetyltransferases in Bacteria.Microbiol Mol Biol Rev. 2020 Apr 15;84(2):e00090-19. doi: 10.1128/MMBR.00090-19. Print 2020 May 20. Microbiol Mol Biol Rev. 2020. PMID: 32295819 Free PMC article. Review.
-
Crystal structure of an aminoglycoside 6'-N-acetyltransferase: defining the GCN5-related N-acetyltransferase superfamily fold.Structure. 1999 May;7(5):497-507. doi: 10.1016/s0969-2126(99)80066-5. Structure. 1999. PMID: 10378269
-
Insights into the specificity of lysine acetyltransferases.J Biol Chem. 2014 Dec 26;289(52):36249-62. doi: 10.1074/jbc.M114.613901. Epub 2014 Nov 7. J Biol Chem. 2014. PMID: 25381442 Free PMC article.
-
Structure and functions of the GNAT superfamily of acetyltransferases.Arch Biochem Biophys. 2005 Jan 1;433(1):212-26. doi: 10.1016/j.abb.2004.09.003. Arch Biochem Biophys. 2005. PMID: 15581578 Review.
Cited by
-
Acetylome analysis of lysine acetylation in the plant pathogenic bacterium Brenneria nigrifluens.Microbiologyopen. 2020 Jan;9(1):e00952. doi: 10.1002/mbo3.952. Epub 2019 Nov 2. Microbiologyopen. 2020. PMID: 31677250 Free PMC article.
-
Freshwater viral metagenome reveals novel and functional phage-borne antibiotic resistance genes.Microbiome. 2020 Jun 1;8(1):75. doi: 10.1186/s40168-020-00863-4. Microbiome. 2020. PMID: 32482165 Free PMC article.
-
Spotlight on Plant Bromodomain Proteins.Biology (Basel). 2023 Aug 2;12(8):1076. doi: 10.3390/biology12081076. Biology (Basel). 2023. PMID: 37626962 Free PMC article. Review.
-
Encystment Induces Down-Regulation of an Acetyltransferase-Like Gene in Acanthamoeba castellanii.Pathogens. 2020 Apr 26;9(5):321. doi: 10.3390/pathogens9050321. Pathogens. 2020. PMID: 32357498 Free PMC article.
-
Arabidopsis thaliana exudates induce growth and proteomic changes in Gluconacetobacter diazotrophicus.PeerJ. 2020 Jul 28;8:e9600. doi: 10.7717/peerj.9600. eCollection 2020. PeerJ. 2020. PMID: 33240578 Free PMC article.
References
-
- Okamoto S, Suzuki Y. Chloramphenicol-, dihydrostreptomycin-, and kanamycin-inactivating enzymes from multiple drug-resistant Escherichia coli carrying episome ‘R’. Nature. 1965;208:1301–1303. - PubMed
-
- Brownell JE, Zhou J, Ranalli T, Kobayashi R, Edmondson DG, Roth SY, Allis CD. Tetrahymena histone acetyltransferase A: a homolog to yeast Gcn5p linking histone acetylation to gene activation. Cell. 1996;84:843–851. - PubMed
-
- Parthun MR, Widom J, Gottschling DE. The major cytoplasmic histone acetyltransferase in yeast: links to chromatin replication and histone metabolism. Cell. 1996;87:85–94. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

