A phase I study for intravenous autologous mesenchymal stromal cell administration to patients with severe emphysema
- PMID: 26819296
- PMCID: PMC4888332
- DOI: 10.1093/qjmed/hcw001
A phase I study for intravenous autologous mesenchymal stromal cell administration to patients with severe emphysema
Abstract
Background: Mesenchymal stromal cells (MSCs) may reduce inflammation and promote tissue repair in pulmonary emphysema.
Aim: To study the safety and feasibility of bone marrow-derived autologous (BM-) MSC intravenous administration to patients with severe emphysema.
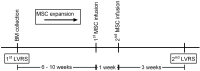
Design: A phase I, prospective open-label study registered at ClinicalTrials.gov as NCT01306513 Eligible patients had lung volume reduction surgery (LVRS) on two separate occasions. During the first LVRS bone marrow was collected, from which MSCs were isolated and expanded ex vivo After 8 weeks, patients received two autologous MSC infusions 1 week apart, followed by the second LVRS procedure at 3 weeks after the second BM-MSC infusion.
Methods: Up to 3 weeks after the last MSC infusion adverse events were recorded. Using immunohistochemistry and qPCR for analysis of cell and proliferation markers, emphysematous lung tissue obtained during the first surgery was compared with lung tissue obtained after the second surgical session to assess BM-MSC effects.
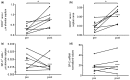
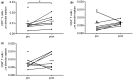
Results: From 10 included patients three were excluded: two did not receive MSCs due to insufficient MSC culture expansion, and one had no second surgery. No adverse events related to MSC infusions occurred and lung tissue showed no fibrotic responses. After LVRS and MSC infusions alveolar septa showed a 3-fold increased expression of the endothelial marker CD31 (P = 0.016).
Conclusions: Autologous MSC treatment in severe emphysema is feasible and safe. The increase in CD31 expression after LVRS and MSC treatment suggests responsiveness of microvascular endothelial cells in the most severely affected parts of the lung.
© The Author 2016. Published by Oxford University Press on behalf of the Association of Physicians. All rights reserved. For Permissions, please email: journals.permissions@oup.com.
Figures

References
-
- Pittenger MF, Mackay AM, Beck SC, Jaiswal RK, Douglas R, Mosca JD, et al. Multilineage potential of adult human mesenchymal stem cells. Science 1999; 284:143–7. - PubMed
-
- Bernardo ME, Fibbe WE. Mesenchymal stromal cells: sensors and switchers of inflammation. Cell Stem Cell 13:392–402. - PubMed
-
- Petrella F, Acocella F, Barberis M, Bellomi M, Brizzola S, Donghi S, et al. Airway fistula closure after stem-cell infusion. N Engl J Med 2014; 372:96–7. - PubMed
-
- Le Blanc K, Frassoni F, Ball L, Locatelli F, Roelofs H, Lewis I, et al. Mesenchymal stem cells for treatment of steroid-resistant, severe, acute graft-versus-host disease: a phase II study. Lancet 2008; 371:1579–86. - PubMed
-
- Molendijk I, Bonsing BA, Roelofs H, Peeters KC, Wasser MN, Dijkstra G, et al. Allogeneic bone marrow-derived mesenchymal stromal cells promote healing of refractory perianal fistulas in patients with crohn's disease. Gastroenterology 2015; 149:918–27. - PubMed
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

