Analysis of the time-to-onset of osteonecrosis of jaw with bisphosphonate treatment using the data from a spontaneous reporting system of adverse drug events
- PMID: 26819745
- PMCID: PMC4728763
- DOI: 10.1186/s40780-015-0035-2
Analysis of the time-to-onset of osteonecrosis of jaw with bisphosphonate treatment using the data from a spontaneous reporting system of adverse drug events
Abstract
Background: Bisphosphonates (BPs) are potent antiresorptive agents used to treat osteoporosis and the complications associated with malignant bone metastasis. The aim of this study was to evaluate the incidence of bisphosphonate-related osteonecrosis of the jaw (BRONJ) using the Japanese Adverse Drug Event Report (JADER) database. In particular, we focused on the time-to-onset profile of BRONJ.
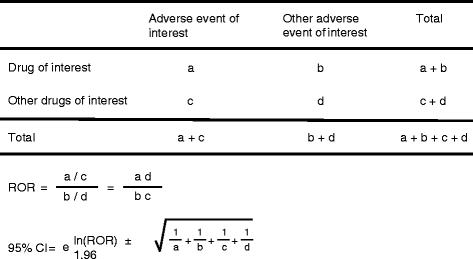
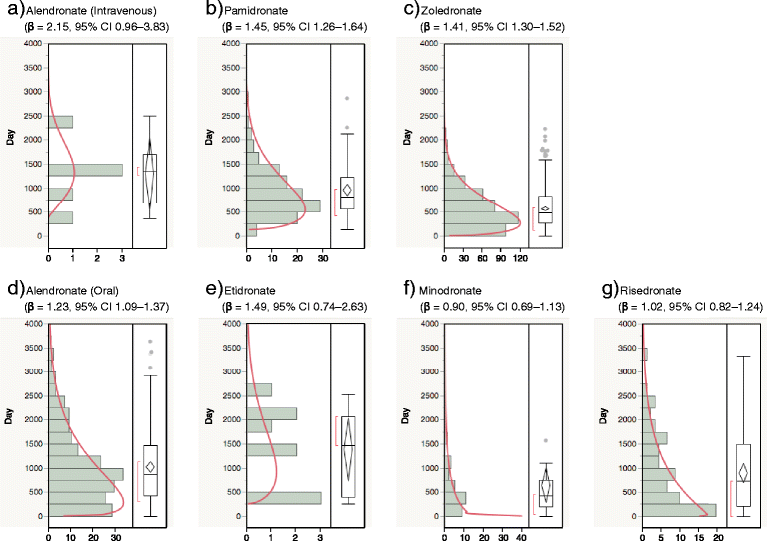
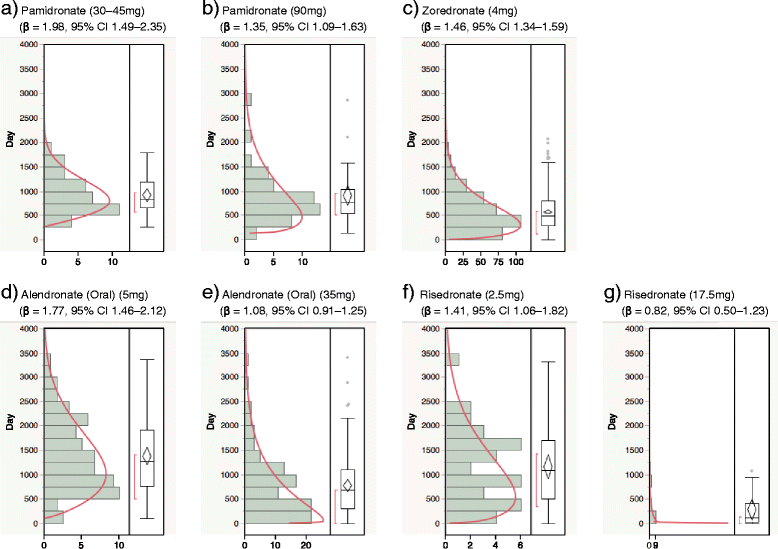
Findings: We analyzed reports of BRONJ in the JADER database and calculated the reporting odds ratio (ROR) of BPs potentially associated with BRONJ. We applied the weibull shape parameter to time-to-event data in JADER. The drugs selected for this investigation were seven BPs approved in Japan (alendronate [intraveneous, I.V.], pamidronate, and zoledronate as I.V. BPs; and alendronate (oral), etidronate, minodronate, and risedronate as oral BPs). We analyzed reports of BRONJ events associated with BPs in the JADER database from April 2004 to November 2014. The median value of BRONJ cases caused by alendronate (I.V.), pamidronate, zoledronate, alendronate (oral), etidronate, minodronate, and risedronate were 1342, 812, 486, 863, 1461, 432, and 730 days, respectively. The lower 95 % confidence interval of the Weibull-shape parameter β for I.V. BPs (pamidronate and zoledronate) exceeded 1. The risk of BRONJ with I.V. BPs increased over time.
Conclusion: Thus, the incidence of BRONJ with BP treatment should be closely monitored for a 3-year period. Further studies are required to draw conclusions, and we believe that this information about BRONJ induced by BPs will prove beneficial to patients and pharmacists.
Keywords: Bisphosphonate; Japanese adverse drug event report database; Osteonecrosis of jaw.
Figures
References
-
- Ruggiero SL, Dodson TB, Fantasia J, Goodday R, Aghaloo T, Mehrotra B, et al. American Association of Oral and Maxillofacial Surgeons. American Association of Oral and Maxillofacial Surgeons position paper on medication-related osteonecrosis of the jaw −2014 update. J Oral Maxillofac Surg. 2014;72:1938–56. doi: 10.1016/j.joms.2014.04.031. - DOI - PubMed
-
- Mariotti A. Bisphosphonates and osteonecrosis of the jaws. J Dent Educ. 2008;72:919–29. - PubMed
-
- Umetsu R, Nishibata Y, Abe J, Suzuki Y, Hara H, Nagasawa H, et al. Evaluation of the association between the use of oral anti-hyperglycemic agents and hypoglycemia in Japan by data mining of the Japanese Adverse Drug Event Report (JADER) database. Yakugaku Zasshi. 2014;134:299–304. doi: 10.1248/yakushi.13-00225. - DOI - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

