Effective Sample Preparations in Imaging Mass Spectrometry
- PMID: 26819901
- PMCID: PMC4542606
- DOI: 10.5702/massspectrometry.S0029
Effective Sample Preparations in Imaging Mass Spectrometry
Abstract
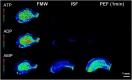
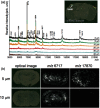
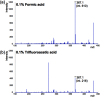
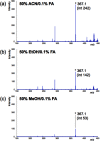
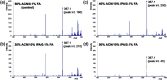
Matrix-assisted laser desorption/ionization (MALDI) imaging mass spectrometry (IMS) can be used to visualize the distribution of biomolecules (proteins, peptides, metabolites) and drugs on tissue surfaces. In MALDI-IMS, sample preparation is crucial for successful results. A variety of conditions, such as tissue sampling methods, tissue thickness and matrix application procedure can have an impact on the results. In this review, we summarize each sample preparation step in an orderly sequence with practical examples. In addition, we discuss the importance of the organic solvent used in the matrix solution. The composition of the organic solvent used in the matrix solution is critical for achieving a high sensitivity in this procedure.
Keywords: adenylates; anti-cancer agents; focused-microwave irradiation; imaging mass spectrometry; sample preparation.
Figures



Similar articles
-
Matrix-assisted laser desorption/ionization and nanoparticle-based imaging mass spectrometry for small metabolites: a practical protocol.Methods Mol Biol. 2010;656:173-95. doi: 10.1007/978-1-60761-746-4_10. Methods Mol Biol. 2010. PMID: 20680591
-
Imaging mass spectrometry: principle and application.Biophys Rev. 2009 Sep;1(3):131. doi: 10.1007/s12551-009-0015-6. Epub 2009 Sep 9. Biophys Rev. 2009. PMID: 28509996 Free PMC article. Review.
-
Utility of imaging mass spectrometry (IMS) by matrix-assisted laser desorption ionization (MALDI) on an ion trap mass spectrometer in the analysis of drugs and metabolites in biological tissues.J Pharmacol Toxicol Methods. 2007 May-Jun;55(3):279-88. doi: 10.1016/j.vascn.2006.11.004. Epub 2006 Dec 5. J Pharmacol Toxicol Methods. 2007. PMID: 17222568
-
MALDI (matrix assisted laser desorption ionization) Imaging Mass Spectrometry (IMS) of skin: Aspects of sample preparation.Talanta. 2017 Nov 1;174:325-335. doi: 10.1016/j.talanta.2017.06.018. Epub 2017 Jun 10. Talanta. 2017. PMID: 28738588
-
MALDI imaging mass spectrometry and analysis of endogenous peptides.Expert Rev Proteomics. 2013 Aug;10(4):381-8. doi: 10.1586/14789450.2013.814939. Expert Rev Proteomics. 2013. PMID: 23992420 Review.
Cited by
-
Advances in mass spectrometry imaging for spatial cancer metabolomics.Mass Spectrom Rev. 2024 Mar-Apr;43(2):235-268. doi: 10.1002/mas.21804. Epub 2022 Sep 6. Mass Spectrom Rev. 2024. PMID: 36065601 Free PMC article. Review.
-
Decellularization and Enzymatic Digestion Methods to Enhance ECM Protein Detection via MALDI-MS Imaging.Anal Chem. 2025 Jan 14;97(1):886-893. doi: 10.1021/acs.analchem.4c05562. Epub 2025 Jan 3. Anal Chem. 2025. PMID: 39753373
-
Branched-Chain Amino Acid Catabolism Promotes Ovarian Cancer Cell Proliferation via Phosphorylation of mTOR.Cancer Res Commun. 2025 Apr 1;5(4):569-579. doi: 10.1158/2767-9764.CRC-24-0532. Cancer Res Commun. 2025. PMID: 40066850 Free PMC article.
-
Sample preparation of formalin-fixed paraffin-embedded tissue sections for MALDI-mass spectrometry imaging.Anal Bioanal Chem. 2020 Feb;412(6):1263-1275. doi: 10.1007/s00216-019-02296-x. Epub 2020 Jan 28. Anal Bioanal Chem. 2020. PMID: 31989198 Free PMC article.
-
Applications of spatially resolved omics in the field of endocrine tumors.Front Endocrinol (Lausanne). 2023 Jan 10;13:993081. doi: 10.3389/fendo.2022.993081. eCollection 2022. Front Endocrinol (Lausanne). 2023. PMID: 36704039 Free PMC article. Review.
References
-
- 3) N. Goto-Inoue, M. Setou, N. Zaima. Visualization of spatial distribution of gamma-aminobutyric acid in eggplant (Solanum melongena) by matrix-assisted laser desorption/ionization imaging mass spectrometry. Anal. Sci. 26: 821–825, 2010. - PubMed
-
- 4) K. Nagashima, A. N. Krot, H. Yurimoto. Stardust silicates from primitive meteorites. Nature 428: 921–924, 2004. - PubMed
-
- 5) S. Rauser, C. Marquardt, B. Balluff, S. Deininger, C. Albers, E. Belau, R. Hartmer, D. Suckau, K. Specht, M. P. Ebert, M. Schmitt, M. Aubele, H. Hoefler, A. Walch. Classification of HER2 receptor status in breast cancer tissues by MALDI imaging mass spectrometry. J. Proteome Res. 9: 1854–1863, 2010. - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources

