Imidazole Alkaloids from the South China Sea Sponge Pericharax heteroraphis and Their Cytotoxic and Antiviral Activities
- PMID: 26821008
- PMCID: PMC6274532
- DOI: 10.3390/molecules21020150
Imidazole Alkaloids from the South China Sea Sponge Pericharax heteroraphis and Their Cytotoxic and Antiviral Activities
Abstract
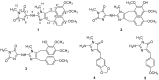
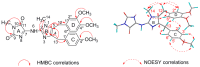
Marine sponges continue to serve as a rich source of alkaloids possessing interesting biological activities and often exhibiting unique structural frameworks. In the current study, chemical investigation on the marine sponge Pericharax heteroraphis collected from the South China Sea yielded one new imidazole alkaloid named naamidine J (1) along with four known ones (2-5). Their structures were established by extensive spectroscopic methods and comparison of their data with those of the related known compounds. All the isolates possessed a central 2-aminoimidazole ring, substituted by one or two functionalized benzyl groups in some combination of the C4 and C5 positions. The cytotoxicities against selected HL-60, HeLa, A549 and K562 tumor cell lines and anti-H1N1 (Influenza a virus (IAV)) activity for the isolates were evaluated. Compounds 1 and 2 exhibited cytotoxicities against the K562 cell line with IC50 values of 11.3 and 9.4 μM, respectively. Compound 5 exhibited weak anti-H1N1 (influenza a virus, IAV) activity with an inhibition ratio of 33%.
Keywords: 2-aminoimidazole alkaloids; Pericharax heteroraphis; anti-H1N1 activity; cytotoxicities; sponge.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
Similar articles
-
Polyhydroxylated steroids from the South China Sea soft coral Sarcophyton sp. and their cytotoxic and antiviral activities.Mar Drugs. 2013 Dec 2;11(12):4788-98. doi: 10.3390/md11124788. Mar Drugs. 2013. PMID: 24317470 Free PMC article.
-
Imidazole Alkaloids and Their Zinc Complexes from the Calcareous Marine Sponge Leucetta chagosensis.J Nat Prod. 2018 Apr 27;81(4):894-900. doi: 10.1021/acs.jnatprod.7b01006. Epub 2018 Apr 12. J Nat Prod. 2018. PMID: 29648818
-
Leucanone A and naamine J, glycerol ether lipid and imidazole alkaloid from the marine sponge Leucandra sp.J Asian Nat Prod Res. 2017 Jul;19(7):691-696. doi: 10.1080/10286020.2016.1240171. Epub 2016 Oct 19. J Asian Nat Prod Res. 2017. PMID: 27756152
-
Review on marine sponge alkaloid, aaptamine: A potential antibacterial and anticancer drug.Chem Biol Drug Des. 2022 Jan;99(1):103-110. doi: 10.1111/cbdd.13932. Epub 2021 Aug 22. Chem Biol Drug Des. 2022. PMID: 34331335 Review.
-
Structure and synthesis of 2-aminoimidazole alkaloids from Leucetta and Clathrina sponges.Nat Prod Rep. 2011 Mar;28(3):511-28. doi: 10.1039/c0np00001a. Epub 2010 Oct 28. Nat Prod Rep. 2011. PMID: 20981389 Review.
Cited by
-
Chemoproteomics of Marine Natural Product Naamidine J Unveils CSE1L as a Therapeutic Target in Acute Lung Injury.J Am Chem Soc. 2024 Oct 16;146(41):28384-28397. doi: 10.1021/jacs.4c09695. Epub 2024 Sep 26. J Am Chem Soc. 2024. PMID: 39324953 Free PMC article.
-
Efficient Total Synthesis of Lissodendrin B, 2-Aminoimidazole Marine Alkaloids Isolated from Lissodendoryx (Acanthodoryx) Fibrosa.Mar Drugs. 2019 Dec 31;18(1):36. doi: 10.3390/md18010036. Mar Drugs. 2019. PMID: 31906213 Free PMC article.
-
Design, Synthesis, and Biological Evaluation of Marine Lissodendrins B Analogues as Modulators of ABCB1-Mediated Multidrug Resistance.Mar Drugs. 2023 May 20;21(5):314. doi: 10.3390/md21050314. Mar Drugs. 2023. PMID: 37233508 Free PMC article.
-
Natural Product-Derived Phytochemicals for Influenza A Virus (H1N1) Prevention and Treatment.Molecules. 2024 May 17;29(10):2371. doi: 10.3390/molecules29102371. Molecules. 2024. PMID: 38792236 Free PMC article. Review.
-
An Anti-Inflammatory 2,4-Cyclized-3,4-Secospongian Diterpenoid and Furanoterpene-Related Metabolites of a Marine Sponge Spongia sp. from the Red Sea.Mar Drugs. 2021 Jan 16;19(1):38. doi: 10.3390/md19010038. Mar Drugs. 2021. PMID: 33467112 Free PMC article.
References
-
- Tsukamoto S., Kawabata T., Kato H., Ohta T., Rotinsulu H., Mangindaan R.E.P., van Soest R.W.M., Ukai K., Kobayashi H., Namikoshi M. Naamidines H and I, Cytotoxic Imidazole Alkaloids from the Indonesian Marine Sponge Leucetta chagosensis. J. Nat. Prod. 2007;70:1658–1660. doi: 10.1021/np070246i. - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous