Daclatasvir, sofosbuvir, and ribavirin for hepatitis C virus genotype 3 and advanced liver disease: A randomized phase III study (ALLY-3+)
- PMID: 26822022
- PMCID: PMC5069621
- DOI: 10.1002/hep.28473
Daclatasvir, sofosbuvir, and ribavirin for hepatitis C virus genotype 3 and advanced liver disease: A randomized phase III study (ALLY-3+)
Abstract
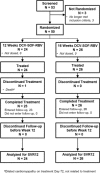
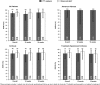
Patients with hepatitis C virus (HCV) genotype 3 infection, especially those with advanced liver disease, are a challenging population in urgent need of optimally effective therapies. The combination of daclatasvir (DCV; pangenotypic nonstructural protein 5A inhibitor) and sofosbuvir (SOF; nucleotide nonstructural protein 5B inhibitor) for 12 weeks previously showed high efficacy (96%) in noncirrhotic genotype 3 infection. The phase III ALLY-3+ study (N = 50) evaluated DCV-SOF with ribavirin (RBV) in treatment-naïve (n = 13) or treatment-experienced (n = 37) genotype 3-infected patients with advanced fibrosis (n = 14) or compensated cirrhosis (n = 36). Patients were randomized 1:1 to receive open-label DCV-SOF (60 + 400 mg daily) with weight-based RBV for 12 or 16 weeks. The primary endpoint was sustained virological response at post-treatment week 12 (SVR12). SVR12 (intention-to-treat) was 90% overall (45 of 50): 88% (21 of 24) in the 12-week (91% observed) and 92% (24 of 26) in the 16-week group. All patients with advanced fibrosis achieved SVR12. SVR12 in patients with cirrhosis was 86% overall (31 of 36): 83% (15 of 18) in the 12-week (88% observed) and 89% (16 of 18) in the 16-week group; for treatment-experienced patients with cirrhosis, these values were 87% (26 of 30), 88% (14 of 16; 93% observed), and 86% (12 of 14), respectively. One patient (12-week group) did not enter post-treatment follow-up (death unrelated to treatment). There were 4 relapses (2 per group) and no virological breakthroughs. The most common adverse events (AEs) were insomnia, fatigue, and headache. There were no discontinuations for AEs and no treatment-related serious AEs.
Conclusion: The all-oral regimen of DCV-SOF-RBV was well tolerated and resulted in high and similar SVR12 after 12 or 16 weeks of treatment among genotype 3-infected patients with advanced liver disease, irrespective of past HCV treatment experience.
© 2016 The Authors. HEPATOLOGY published by Wiley Periodicals, Inc., on behalf of the American Association for the Study of Liver Diseases.
Figures
References
-
- Shepard CW, Finelli L, Alter MJ. Global epidemiology of hepatitis C virus infection. Lancet Infect Dis 2005;5:558‐567. - PubMed
-
- Stamouli M, Panagiotou I, Kairis D, Michopoulou A, Skliris A, Totos G. Genotype distribution in chronic hepatitis C patients in Greece. Clin Lab 2012;58:173‐176. - PubMed
-
- Chlabicz S, Flisiak R, Kowalczuk O, Grzeszczuk A, Pytel‐Krolczuk B, Prokopowicz D, et al. Changing HCV genotypes distribution in Poland‐relation to source and time of infection. J Clin Virol 2008;42:156‐159. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous