Beclin1 and HMGB1 ameliorate the α-synuclein-mediated autophagy inhibition in PC12 cells
- PMID: 26822891
- PMCID: PMC4731928
- DOI: 10.1186/s13000-016-0459-5
Beclin1 and HMGB1 ameliorate the α-synuclein-mediated autophagy inhibition in PC12 cells
Abstract
Background: Aberrant α-synuclein aggregation due to the deficiency of ubiquitin-proteasome or of autophagy characterizes the parkinson disease (PD). High mobility group box 1 (HMGB1) is a novel stress sensor to mediate the persistent neuro-inflammation and the consequent progressive neurodegeneration, via controlling the cellular autophagy/apoptosis checkpoint during inflammation. Moreover, HMGB1 has been recently indicated to involve in the autophagic degradation of α-synuclein.
Methods: In the current study, we investigated the influence of the overexpressed α-synuclein of wild type (wt) or mutant type (A53T and A30P, mt) on the cytosolic levels of HMGB1 and Beclin1 and on the starvation-induced autophagy in pheochromocytoma PC12 cells. And then we explored the overexpression of HMGB1 or of Beclin1 on the α-synuclein degradation and on the autophagy in the α-synuclein-overexpressed PC12 cells.
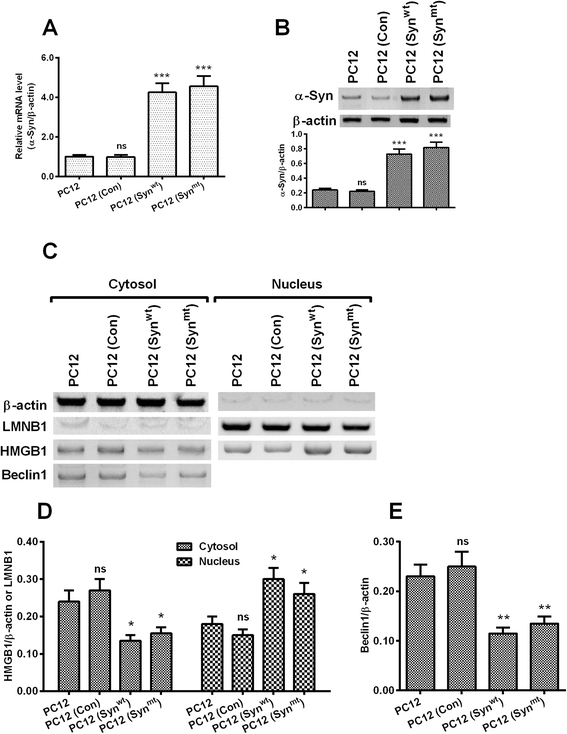
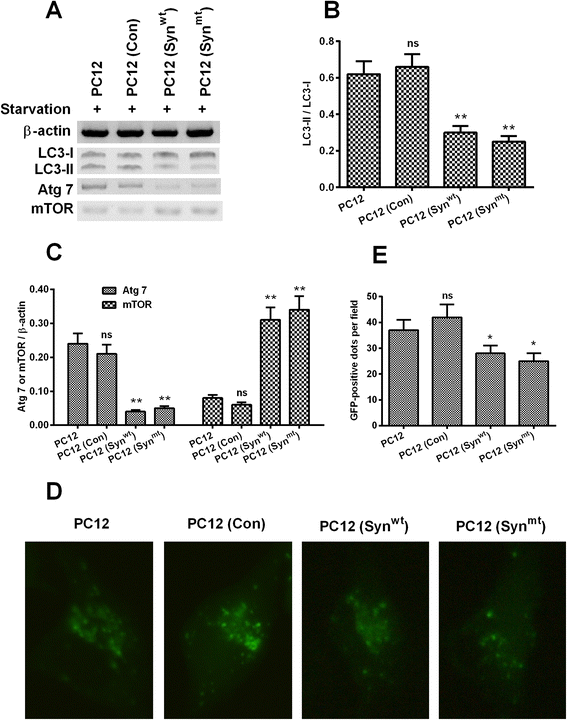
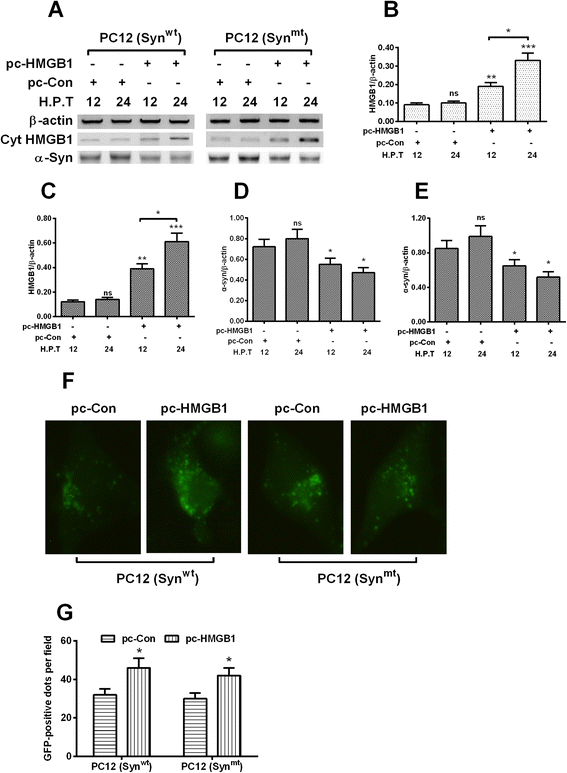
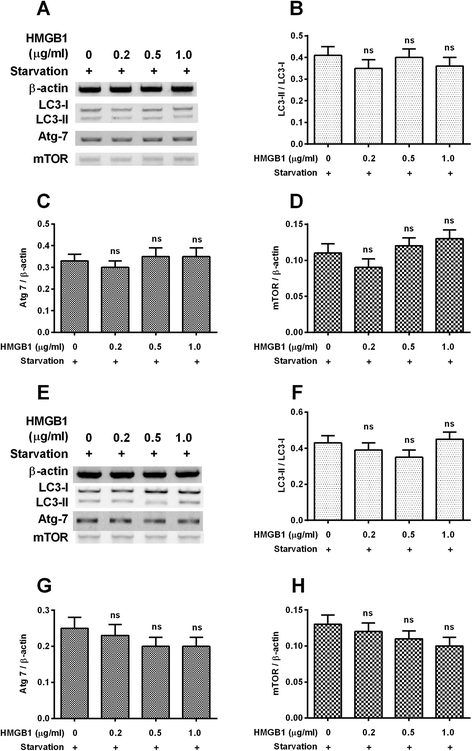
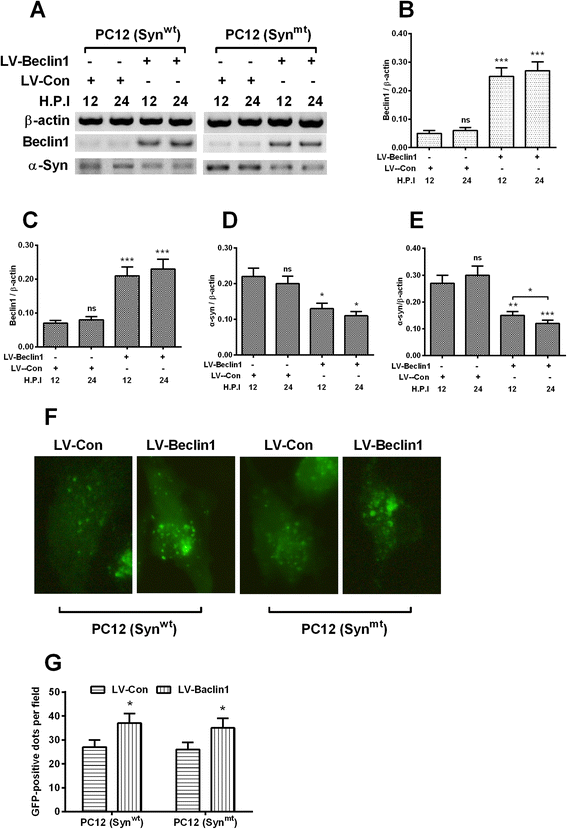
Results: It was demonstrated that α-synuclein overexpression inhibited the trans-location of HMGB1 from nucleus to cytosol and reduced the cytosolic level of Beclin1 in PC12 cells, and inhibited the starvation-induced autophagy via downregulating autophagy-associated markers and via reducing the autophagic vesicles in PC12 cells under starvation. On the other side, the intracellular promotion of either HMGB1 or Beclin1 upregulated the α-synuclein degradation and ameliorated the α-synuclein-mediated autophagy reduction in PC12 cells. However, the exogenous HMGB1 treatment exerted no such regulation in PC12 cells.
Conclusion: In summary, our study confirmed the positive regulation by HMGB1 and Beclin1 on the α-synuclein degradation and on the starvation-induced autophagy in PC12 cells, implying both markers as prominent targets to promote the α-synuclein degradation.
Figures
Similar articles
-
HMGB1 promotes the starvation-induced autophagic degradation of α-synuclein in SH-SY5Y cells Atg 5-dependently.Life Sci. 2018 Jun 1;202:1-10. doi: 10.1016/j.lfs.2018.03.031. Epub 2018 Mar 15. Life Sci. 2018. PMID: 29551576
-
HMGB1 is involved in autophagy inhibition caused by SNCA/α-synuclein overexpression: a process modulated by the natural autophagy inducer corynoxine B.Autophagy. 2014 Jan;10(1):144-54. doi: 10.4161/auto.26751. Epub 2013 Jan 1. Autophagy. 2014. PMID: 24178442 Free PMC article.
-
Corynoxine B ameliorates HMGB1-dependent autophagy dysfunction during manganese exposure in SH-SY5Y human neuroblastoma cells.Food Chem Toxicol. 2019 Feb;124:336-348. doi: 10.1016/j.fct.2018.12.027. Epub 2018 Dec 19. Food Chem Toxicol. 2019. PMID: 30578841
-
Impaired autophagy and APP processing in Alzheimer's disease: The potential role of Beclin 1 interactome.Prog Neurobiol. 2013 Jul-Aug;106-107:33-54. doi: 10.1016/j.pneurobio.2013.06.002. Epub 2013 Jul 1. Prog Neurobiol. 2013. PMID: 23827971 Review.
-
High Mobility Group Box 1 Protein: A Plausible Therapeutic Molecular Target in Parkinson's Disease.Curr Pharm Biotechnol. 2024;25(8):937-943. doi: 10.2174/1389201025666230905092218. Curr Pharm Biotechnol. 2024. PMID: 37670710 Review.
Cited by
-
Targeting α-synuclein for PD Therapeutics: A Pursuit on All Fronts.Biomolecules. 2020 Mar 3;10(3):391. doi: 10.3390/biom10030391. Biomolecules. 2020. PMID: 32138193 Free PMC article. Review.
-
Alpha-Synuclein Contribution to Neuronal and Glial Damage in Parkinson's Disease.Int J Mol Sci. 2023 Dec 26;25(1):360. doi: 10.3390/ijms25010360. Int J Mol Sci. 2023. PMID: 38203531 Free PMC article. Review.
-
Alpha-Synuclein as a Biomarker of Parkinson's Disease: Good, but Not Good Enough.Front Aging Neurosci. 2021 Jul 8;13:702639. doi: 10.3389/fnagi.2021.702639. eCollection 2021. Front Aging Neurosci. 2021. PMID: 34305577 Free PMC article. Review.
-
Autophagy in α-Synucleinopathies-An Overstrained System.Cells. 2021 Nov 12;10(11):3143. doi: 10.3390/cells10113143. Cells. 2021. PMID: 34831366 Free PMC article. Review.
-
Cellular and Molecular Basis of Neurodegeneration in Parkinson Disease.Front Aging Neurosci. 2018 Apr 17;10:109. doi: 10.3389/fnagi.2018.00109. eCollection 2018. Front Aging Neurosci. 2018. PMID: 29719505 Free PMC article. Review.
References
-
- Irizarry MC, Growdon W, Gomez-Isla T, Newell K, George JM, Clayton DF, et al. Nigral and cortical Lewy bodies and dystrophic nigral neurites in Parkinson’s disease and cortical Lewy body disease contain alpha-synuclein immunoreactivity. J Neuropathol Exp Neurol. 1998;57(4):334–337. doi: 10.1097/00005072-199804000-00005. - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

