A new oxygen modification cyclooctaoxygen binds to nucleic acids as sodium crown complex
- PMID: 26825775
- PMCID: PMC4780752
- DOI: 10.1016/j.bbagen.2016.01.022
A new oxygen modification cyclooctaoxygen binds to nucleic acids as sodium crown complex
Abstract
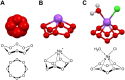
Background: Oxygen exists in two gaseous and six solid allotropic modifications. An additional allotropic modification of oxygen, the cyclooctaoxygen, was predicted to exist in 1990.
Methods: Cyclooctaoxygen sodium was synthesized in vitro from atmospheric oxygen, or catalase effect-generated oxygen, under catalysis of cytosine nucleosides and either ninhydrin or eukaryotic low-molecular weight RNA. Thin-layer chromatographic mobility shift assays were applied on specific nucleic acids and the cyclooctaoxygen sodium complex.
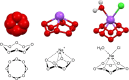
Results: We report the first synthesis and characterization of cyclooctaoxygen as its sodium crown complex, isolated in the form of three cytosine nucleoside hydrochloride complexes. The cationic cyclooctaoxygen sodium complex is shown to bind to nucleic acids (RNA and DNA), to associate with single-stranded DNA and spermine phosphate, and to be essentially non-toxic to cultured mammalian cells at 0.1-1.0mM concentration.
Conclusions: We postulate that cyclooctaoxygen is formed in most eukaryotic cells in vivo from dihydrogen peroxide in a catalase reaction catalyzed by cytidine and RNA. A molecular biological model is deduced for a first epigenetic shell of eukaryotic in vivo DNA. This model incorporates an epigenetic explanation for the interactions of the essential micronutrient selenium (as selenite) with eukaryotic in vivo DNA.
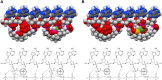
General significance: Since the sperminium phosphate/cyclooctaoxygen sodium complex is calculated to cover the active regions (2.6%) of bovine lymphocyte interphase genome, and 12.4% of murine enterocyte mitotic chromatin, we propose that the sperminium phosphate/cyclooctaoxygen sodium complex coverage of nucleic acids is essential to eukaryotic gene regulation and promoted proto-eukaryotic evolution.
Keywords: Cyclooctaoxygen; DNA; Epigenetics; Oxygen modification; RNA; Selenium.
Copyright © 2016 Elsevier B.V. All rights reserved.
Figures


References
-
- Freiman Yu.A., Jodl H.J. Solid oxygen. Phys. Rep. 2004;401:1–288.
-
- Lundegaard L.F., Weck G., McMahon M.I., Desgreniers S., Loubeyre P. Observation of an O8 molecular lattice in the ε phase of solid oxygen. Nature. 2006;443:201–204. - PubMed
-
- Kim K.S., Jang J.H., Kim S., Mhin B.-J., Schaefer III H.F. Potential new high energy density materials: cyclooctaoxygen O8, including comparisons with the well-known cyclo-S8 molecule. J. Chem. Phys. 1990;92:1887–1892.
-
- Kleinman M.H., Telo J.P., Vieira A.J.S.C., Bohne C., Netto-Ferreira J.C. Transient spectroscopy of ninhydrin. Photochem. Photobiol. 2003;77:10–17. - PubMed
-
- Burger R., Bigler P. DEPTQ: distorsionless enhancement by polarization transfer including the detection of quaternary nuclei. J. Magn. Reson. 1998;135:529–534. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

