Hepcidin-2 in mouse urine as a candidate radiation-responsive molecule
- PMID: 26826199
- PMCID: PMC4795955
- DOI: 10.1093/jrr/rrv098
Hepcidin-2 in mouse urine as a candidate radiation-responsive molecule
Abstract
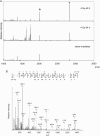
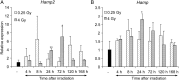
We used high-performance liquid chromatography to separate urine obtained from whole-body gamma-irradiated mice (4 Gy) before analyzing each fraction with matrix-assisted laser desorption/ionization-time-of-flight mass spectrometry to identify radiation-responsive molecules. We identified two candidates: hepcidin antimicrobial peptide 2 (hepcidin-2) and peptide fragments of kidney androgen-regulated protein (KAP). We observed that peak increases of hepcidin-2 in urine were delayed in a dose-dependent manner (1 Gy and above); however, the amount of KAP peptide fragments showed no correlation with radiation dose. In addition, an increase in hepcidin-2 after exposure to relatively low radiation doses (0.25 and 0.5 Gy, respectively) was biphasic (at 8-48 h and 120-168 h, respectively, after irradiation). The increase in hepcidin-2 paralleled an increase in hepcidin-2 gene (Hamp2) mRNA levels in the liver. These results suggest that radiation exposure directly or indirectly induces urinary excretion of hepcidin-2 at least in part by the upregulation of Hamp2 mRNA in the liver.
Keywords: hepcidin-2; mass spectrometry; radiation-responsive molecules; urine.
© The Author 2016. Published by Oxford University Press on behalf of The Japan Radiation Research Society and Japanese Society for Radiation Oncology.
Figures


Similar articles
-
Upregulation of iron regulatory hormone hepcidin by interferon α.J Gastroenterol Hepatol. 2014 Feb;29(2):387-94. doi: 10.1111/jgh.12348. J Gastroenterol Hepatol. 2014. PMID: 23926964
-
Urinary Hepcidin Levels in Iron-Deficient and Iron-Supplemented Piglets Correlate with Hepcidin Hepatic mRNA and Serum Levels and with Body Iron Status.PLoS One. 2015 Aug 31;10(8):e0136695. doi: 10.1371/journal.pone.0136695. eCollection 2015. PLoS One. 2015. PMID: 26323096 Free PMC article.
-
Effects of a single dose of oral iron on hepcidin concentrations in human urine and serum analyzed by a robust LC-MS/MS method.Clin Chim Acta. 2011 Nov 20;412(23-24):2241-7. doi: 10.1016/j.cca.2011.08.014. Epub 2011 Aug 16. Clin Chim Acta. 2011. PMID: 21867695 Free PMC article.
-
Inductively coupled mass spectrometry analysis of biometals in conditional Hamp1 and Hamp1 and Hamp2 transgenic mouse models.Transgenic Res. 2015 Aug;24(4):765-73. doi: 10.1007/s11248-015-9879-3. Epub 2015 Apr 23. Transgenic Res. 2015. PMID: 25904410 Free PMC article.
-
Laboratory use of hepcidin in renal transplant recipients.Biochem Med (Zagreb). 2016;26(1):34-52. doi: 10.11613/BM.2016.003. Biochem Med (Zagreb). 2016. PMID: 26981017 Free PMC article. Review.
Cited by
-
Organ-Specific Effects of Low Dose Radiation Exposure: A Comprehensive Review.Front Genet. 2020 Oct 2;11:566244. doi: 10.3389/fgene.2020.566244. eCollection 2020. Front Genet. 2020. PMID: 33133150 Free PMC article. Review.
-
Antibacterial Activity and Mechanisms of TroHepc2-22, a Derived Peptide of Hepcidin2 from Golden Pompano (Trachinotus ovatus).Int J Mol Sci. 2023 May 25;24(11):9251. doi: 10.3390/ijms24119251. Int J Mol Sci. 2023. PMID: 37298202 Free PMC article.
-
Deposition of Iron in the Bone Marrow of a Murine Model of Hematopoietic Acute Radiation Syndrome.Exp Hematol. 2020 Apr;84:54-66. doi: 10.1016/j.exphem.2020.03.004. Epub 2020 Mar 30. Exp Hematol. 2020. PMID: 32240658 Free PMC article.
-
Analysis of a lectin microarray identifies altered sialylation of mouse serum glycoproteins induced by whole-body radiation exposure.J Radiat Res. 2019 Mar 1;60(2):189-196. doi: 10.1093/jrr/rry100. J Radiat Res. 2019. PMID: 30521038 Free PMC article.
-
Metabolomic screening using ESI-FT MS identifies potential radiation-responsive molecules in mouse urine.J Radiat Res. 2017 May 1;58(3):273-280. doi: 10.1093/jrr/rrw112. J Radiat Res. 2017. PMID: 27974505 Free PMC article.
References
-
- Chen C, Brenner DJ, Brown TR . Identification of urinary biomarkers from X-irradiated mice using NMR spectroscopy . Radiat Res 2011. ; 175 : 622 – 30 . - PubMed
-
- Lanz C, Patterson AD, Slavík J, et al. Radiation metabolomics. 3. Biomarker discovery in the urine of gamma-irradiated rats using a simplified metabolomics protocol of gas chromatography–mass spectrometry combined with random forests machine learning algorithm . Radiat Res 2009. ; 172 : 198 – 212 . - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

