Identification of a binding site of the human immunodeficiency virus envelope protein gp120 to neuronal-specific tubulin
- PMID: 26826352
- PMCID: PMC4828312
- DOI: 10.1111/jnc.13557
Identification of a binding site of the human immunodeficiency virus envelope protein gp120 to neuronal-specific tubulin
Abstract
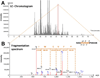
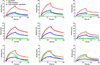
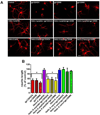
Human immunodeficiency virus-1 (HIV) promotes synaptic simplification and neuronal apoptosis, and causes neurological impairments termed HIV-associated neurological disorders. HIV-associated neurotoxicity may be brought about by acute and chronic mechanisms that still remain to be fully characterized. The HIV envelope glycoprotein gp120 causes neuronal degeneration similar to that observed in HIV-associated neurocognitive disorders subjects. This study was undertaken to discover novel mechanisms of gp120 neurotoxicity that could explain how the envelope protein promotes neurite pruning. Gp120 has been shown to associate with various intracellular organelles as well as microtubules in neurons. We then analyzed lysates of neurons exposed to gp120 with liquid chromatography mass spectrometry for potential protein interactors. We found that one of the proteins interacting with gp120 is tubulin β-3 (TUBB3), a major component of neuronal microtubules. We then tested the hypothesis that gp120 binds to neuronal microtubules. Using surface plasmon resonance, we confirmed that gp120 binds with high affinity to neuronal-specific TUBB3. We have also identified the binding site of gp120 to TUBB3. We then designed a small peptide (Helix-A) that displaced gp120 from binding to TUBB3. To determine whether this peptide could prevent gp120-mediated neurotoxicity, we cross-linked Helix-A to mesoporous silica nanoparticles (Helix-A nano) to enhance the intracellular delivery of the peptide. We then tested the neuroprotective property of Helix-A nano against three strains of gp120 in rat cortical neurons. Helix-A nano prevented gp120-mediated neurite simplification as well as neuronal loss. These data propose that gp120 binding to TUBB3 could be another mechanism of gp120 neurotoxicity. We propose a novel direct mechanism of human immunodeficiency virus neurotoxicity. Our data show that the viral protein gp120 binds to neuronal specific tubulin β-3 and blocks microtubule transport. Displacing gp120 from binding to tubulin by a small peptide prevents gp120-mediated neuronal loss. Our study reveals a novel target for developing adjunct therapies against viral infection that promotes neurocognitive disorders.
Keywords: HAND; Tat; nanoparticles; neurite pruning; neuronal loss; tubulin β-3.
© 2016 International Society for Neurochemistry.
Figures



References
-
- Altar CA, Cai N, Bliven T, Juhasz M, Conner JM, Acheson AL, Lindsay RM, Wiegand SJ. Anterograde transport of brain-derived neurotrophic factor and its role in the brain. Nature. 1997;389:856–860. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

