Accurately Achieving Target Busulfan Exposure in Children and Adolescents With Very Limited Sampling and the BestDose Software
- PMID: 26829600
- PMCID: PMC4864122
- DOI: 10.1097/FTD.0000000000000276
Accurately Achieving Target Busulfan Exposure in Children and Adolescents With Very Limited Sampling and the BestDose Software
Abstract
Background: Busulfan dose adjustment is routinely guided by plasma concentration monitoring using 4-9 blood samples per dose adjustment, but a pharmacometric Bayesian approach could reduce this sample burden.
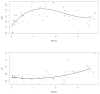
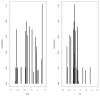
Methods: The authors developed a nonparametric population model with Pmetrics. They used it to simulate optimal initial busulfan dosages, and in a blinded manner, they compared dosage adjustments using the model in the BestDose software to dosage adjustments calculated by noncompartmental estimation of area under the time-concentration curve at a national reference laboratory in a cohort of patients not included in model building.
Results: Mean (range) age of the 53 model-building subjects was 7.8 years (0.2-19.0 years) and weight was 26.5 kg (5.6-78.0 kg), similar to nearly 120 validation subjects. There were 16.7 samples (6-26 samples) per subject to build the model. The BestDose cohort was also diverse: 10.2 years (0.25-18 years) and 46.4 kg (5.2-110.9 kg). Mean bias and imprecision of the 1-compartment model-predicted busulfan concentrations were 0.42% and 9.2%, and were similar in the validation cohorts. Initial dosages to achieve average concentrations of 600-900 ng/mL were 1.1 mg/kg (≤12 kg, 67% in the target range) and 1.0 mg/kg (>12 kg, 76% in the target range). Using all 9 concentrations after dose 1 in the Bayesian estimation of dose requirements, the mean (95% confidence interval) bias of BestDose calculations for the third dose was 0.2% (-2.4% to 2.9%, P = 0.85), compared with the standard noncompartmental method based on 9 concentrations. With 1 optimally timed concentration 15 minutes after the infusion (calculated with the authors' novel MMopt algorithm) bias was -9.2% (-16.7% to -1.5%, P = 0.02). With 2 concentrations at 15 minutes and 4 hours bias was only 1.9% (-0.3% to 4.2%, P = 0.08).
Conclusions: BestDose accurately calculates busulfan intravenous dosage requirements to achieve target plasma exposures in children up to 18 years of age and 110 kg using only 2 blood samples per adjustment compared with 6-9 samples for standard noncompartmental dose calculations.
Conflict of interest statement
None.
Figures
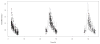
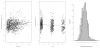

References
-
- Vassal G, Michel G, Espérou H, Gentet JC, Valteau-Couanet D, Doz F, et al. Prospective validation of a novel IV busulfan fixed dosing for paediatric patients to improve therapeutic AUC targeting without drug monitoring. Cancer Chemother Pharmacol. 2008 Jan;61(1):113–23. - PubMed
-
- Schechter T, Finkelstein Y, Doyle J, Verjee Z, Moretti M, Koren G, et al. Pharmacokinetic disposition and clinical outcomes in infants and children receiving intravenous busulfan for allogeneic hematopoietic stem cell transplantation. Biol Blood Marrow Transplant. 2007 Mar;13(3):307–14. - PubMed
-
- Nguyen L, Fuller D, Lennon S, Leger F. IV busulfan in pediatrics: a novel dosing to improve safety/efficacy for hematopoietic progenitor cell transplantation recipients. Bone Marrow Transplant. 2004 May;33(10):979–87. - PubMed
-
- Dix SP, Wingard JR, Mullins RE, Jerkunica I, Davidson TG, Gilmore CE, et al. Association of busulfan area under the curve with veno-occlusive disease following BMT. Bone Marrow Transplant. 1996 Feb;17(2):225–30. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

