Novel flow cytometric approach for the detection of adipocyte subpopulations during adipogenesis
- PMID: 26830859
- PMCID: PMC4808761
- DOI: 10.1194/jlr.D065664
Novel flow cytometric approach for the detection of adipocyte subpopulations during adipogenesis
Abstract
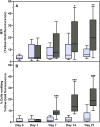
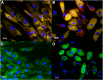
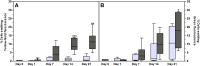
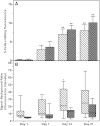
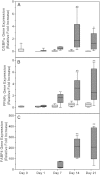
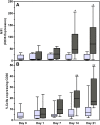
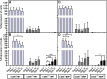
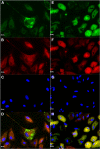
The ability of mesenchymal stromal cells (MSCs) to differentiate into adipocytes provides a cellular model of human origin to study adipogenesis in vitro. One of the major challenges in studying adipogenesis is the lack of tools to identify and monitor the differentiation of various subpopulations within the heterogeneous pool of MSCs. Cluster of differentiation (CD)36 plays an important role in the formation of intracellular lipid droplets, a key characteristic of adipocyte differentiation/maturation. The objective of this study was to develop a reproducible quantitative method to study adipocyte differentiation by comparing two lipophilic dyes [Nile Red (NR) and Bodipy 493/503] in combination with CD36 surface marker staining. We identified a subpopulation of adipose-derived stromal cells that express CD36 at intermediate/high levels and show that combining CD36 cell surface staining with neutral lipid-specific staining allows us to monitor differentiation of adipose-derived stromal cells that express CD36(intermediate/high)during adipocyte differentiation in vitro. The gradual increase of CD36(intermediate/high/)NR(positive)cells during the 21 day adipogenesis induction period correlated with upregulation of adipogenesis-associated gene expression.
Keywords: Bodipy 493/503; Nile Red; adipocyte differentiation; adipose tissue-derived stromal cells; cluster of differentiation 36; fatty/acid binding protein; gene expression; lipid droplet; triglyceride.
Copyright © 2016 by the American Society for Biochemistry and Molecular Biology, Inc.
Figures

Similar articles
-
FABP4 Controls Fat Mass Expandability (Adipocyte Size and Number) through Inhibition of CD36/SR-B2 Signalling.Int J Mol Sci. 2023 Jan 5;24(2):1032. doi: 10.3390/ijms24021032. Int J Mol Sci. 2023. PMID: 36674544 Free PMC article.
-
CD36 Is a Marker of Human Adipocyte Progenitors with Pronounced Adipogenic and Triglyceride Accumulation Potential.Stem Cells. 2017 Jul;35(7):1799-1814. doi: 10.1002/stem.2635. Epub 2017 May 18. Stem Cells. 2017. PMID: 28470788 Clinical Trial.
-
6-gingerol inhibits rosiglitazone-induced adipogenesis in 3T3-L1 adipocytes.Phytother Res. 2014 Feb;28(2):187-92. doi: 10.1002/ptr.4976. Epub 2013 Mar 21. Phytother Res. 2014. PMID: 23519881
-
Fat Cell and Fatty Acid Turnover in Obesity.Adv Exp Med Biol. 2017;960:135-160. doi: 10.1007/978-3-319-48382-5_6. Adv Exp Med Biol. 2017. PMID: 28585198 Review.
-
Review: Epigenetic regulation of adipocyte differentiation and adipogenesis.J Zhejiang Univ Sci B. 2010 Oct;11(10):784-91. doi: 10.1631/jzus.B0900401. J Zhejiang Univ Sci B. 2010. PMID: 20872986 Free PMC article. Review.
Cited by
-
Live Simultaneous Monitoring of Mineral Deposition and Lipid Accumulation in Differentiating Stem Cells.Biomimetics (Basel). 2019 Jul 10;4(3):48. doi: 10.3390/biomimetics4030048. Biomimetics (Basel). 2019. PMID: 31295946 Free PMC article.
-
Quantification of Adipogenesis Using Flow Cytometry.Methods Mol Biol. 2025;2938:13-19. doi: 10.1007/978-1-0716-4607-6_2. Methods Mol Biol. 2025. PMID: 40445275
-
Adipocyte-Based Cell Therapy in Oncology: The Role of Cancer-Associated Adipocytes and Their Reinterpretation as Delivery Platforms.Pharmaceutics. 2020 Apr 28;12(5):402. doi: 10.3390/pharmaceutics12050402. Pharmaceutics. 2020. PMID: 32354024 Free PMC article. Review.
-
Chemically Defined Xeno- and Serum-Free Cell Culture Medium to Grow Human Adipose Stem Cells.Cells. 2021 Feb 22;10(2):466. doi: 10.3390/cells10020466. Cells. 2021. PMID: 33671568 Free PMC article.
-
Single-cell RNA-seq highlights heterogeneity in human primary Wharton's jelly mesenchymal stem/stromal cells cultured in vitro.Stem Cell Res Ther. 2020 Apr 6;11(1):149. doi: 10.1186/s13287-020-01660-4. Stem Cell Res Ther. 2020. PMID: 32252818 Free PMC article.
References
-
- Mosna F., Sensebé L., and Krampera M.. 2010. Human bone marrow and adipose tissue mesenchymal stem cells: a user’s guide. Stem Cells Dev. 19: 1449–1470. - PubMed
-
- Bourin P., Bunnell B. A., Casteilla L., Dominici M., Katz A. J., March K. L., Redl H., Rubin J. P., Yoshimura K., and Gimble J. M.. 2013. Stromal cells from the adipose tissue-derived stromal vascular fraction and culture expanded adipose tissue-derived stromal/stem cells: a joint statement of the International Federation for Adipose Therapeutics and Science (IFATS) and the International Society for Cellular Therapy (ISCT). Cytotherapy. 15: 641–648. - PMC - PubMed
-
- Vishnubalaji R., Al-Nbaheen M., Kadalmani B., Aldahmash A., and Ramesh T.. 2012. Comparative investigation of the differentiation capability of bone-marrow- and adipose-derived mesenchymal stem cells by qualitative and quantitative analysis. Cell Tissue Res. 347: 419–427. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

