OligoG CF-5/20 Disruption of Mucoid Pseudomonas aeruginosa Biofilm in a Murine Lung Infection Model
- PMID: 26833153
- PMCID: PMC4862494
- DOI: 10.1128/AAC.01721-15
OligoG CF-5/20 Disruption of Mucoid Pseudomonas aeruginosa Biofilm in a Murine Lung Infection Model
Abstract
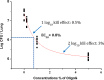
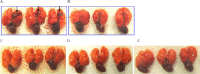
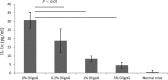
Biofilm growth is a universal survival strategy for bacteria, providing an effective and resilient approach for survival in an otherwise hostile environment. In the context of an infection, a biofilm provides resistance and tolerance to host immune defenses and antibiotics, allowing the biofilm population to survive and thrive under conditions that would destroy their planktonic counterparts. Therefore, the disruption of the biofilm is a key step in eradicating persistent bacterial infections, as seen in many types of chronic disease. In these studies, we used both in vitro minimum biofilm eradication concentration (MBEC) assays and an in vivo model of chronic biofilm infection to demonstrate the biofilm-disrupting effects of an alginate oligomer, OligoG CF-5/20. Biofilm infections were established in mice by tracheal instillation of a mucoid clinical isolate of Pseudomonas aeruginosa embedded in alginate polymer beads. The disruption of the biofilm by OligoG CF-5/20 was observed in a dose-dependent manner over 24 h, with up to a 2.5-log reduction in CFU in the infected mouse lungs. Furthermore, in vitro assays showed that 5% OligoG CF-5/20 significantly reduced the MBEC for colistin from 512 μg/ml to 4 μg/ml after 8 h. These findings support the potential for OligoG CF-5/20 as a biofilm disruption agent which may have clinical value in reducing the microbial burden in chronic biofilm infections.
Copyright © 2016, American Society for Microbiology. All Rights Reserved.
Figures
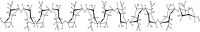



Similar articles
-
A Low-Molecular-Weight Alginate Oligosaccharide Disrupts Pseudomonal Microcolony Formation and Enhances Antibiotic Effectiveness.Antimicrob Agents Chemother. 2017 Aug 24;61(9):e00762-17. doi: 10.1128/AAC.00762-17. Print 2017 Sep. Antimicrob Agents Chemother. 2017. PMID: 28630204 Free PMC article.
-
Phenotypic and Genotypic Adaptations in Pseudomonas aeruginosa Biofilms following Long-Term Exposure to an Alginate Oligomer Therapy.mSphere. 2021 Jan 20;6(1):e01216-20. doi: 10.1128/mSphere.01216-20. mSphere. 2021. PMID: 33472983 Free PMC article.
-
Optimization of colistin dosing regimen for cystic fibrosis patients with chronic Pseudomonas aeruginosa biofilm lung infections.Pediatr Pulmonol. 2019 May;54(5):575-580. doi: 10.1002/ppul.24269. Epub 2019 Feb 25. Pediatr Pulmonol. 2019. PMID: 30803159
-
Pseudomonas aeruginosa chromosomal beta-lactamase in patients with cystic fibrosis and chronic lung infection. Mechanism of antibiotic resistance and target of the humoral immune response.APMIS Suppl. 2003;(116):1-47. APMIS Suppl. 2003. PMID: 14692154 Review.
-
Pseudomonas aeruginosa biofilms in cystic fibrosis.Future Microbiol. 2010 Nov;5(11):1663-74. doi: 10.2217/fmb.10.125. Future Microbiol. 2010. PMID: 21133688 Review.
Cited by
-
The use of combination therapy for the improvement of colistin activity against bacterial biofilm.Braz J Microbiol. 2024 Mar;55(1):411-427. doi: 10.1007/s42770-023-01189-7. Epub 2023 Nov 30. Braz J Microbiol. 2024. PMID: 38030866 Free PMC article. Review.
-
Bioarchitectural Design of Bioactive Biopolymers: Structure-Function Paradigm for Diabetic Wound Healing.Biomimetics (Basel). 2024 May 4;9(5):275. doi: 10.3390/biomimetics9050275. Biomimetics (Basel). 2024. PMID: 38786486 Free PMC article. Review.
-
Recent Advances in Alginate Lyase Engineering for Efficient Conversion of Alginate to Value-Added Products.Microb Biotechnol. 2025 May;18(5):e70150. doi: 10.1111/1751-7915.70150. Microb Biotechnol. 2025. PMID: 40293191 Free PMC article. Review.
-
Alginate Oligosaccharide and Gut Microbiota: Exploring the Key to Health.Nutrients. 2025 Jun 11;17(12):1977. doi: 10.3390/nu17121977. Nutrients. 2025. PMID: 40573088 Free PMC article. Review.
-
Combination treatment to improve mucociliary transport of Pseudomonas aeruginosa biofilms.PLoS One. 2024 Feb 23;19(2):e0294120. doi: 10.1371/journal.pone.0294120. eCollection 2024. PLoS One. 2024. PMID: 38394229 Free PMC article.
References
-
- Ciofu O, Fussing V, Bagge N, Koch C, Høiby N. 2001. Characterization of paired mucoid/non-mucoid Pseudomonas aeruginosa isolates from Danish cystic fibrosis patients: antibiotic resistance, β-lactamase activity and RiboPrinting. J Antimicrob Chemother 48:391–396. doi:10.1093/jac/48.3.391. - DOI - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

