Immunotherapy of cancer: from monoclonal to oligoclonal cocktails of anti-cancer antibodies: IUPHAR Review 18
- PMID: 26833433
- PMCID: PMC4831314
- DOI: 10.1111/bph.13450
Immunotherapy of cancer: from monoclonal to oligoclonal cocktails of anti-cancer antibodies: IUPHAR Review 18
Abstract
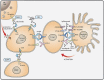
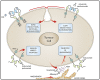
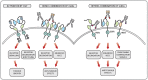
Antibody-based therapy of cancer employs monoclonal antibodies (mAbs) specific to soluble ligands, membrane antigens of T-lymphocytes or proteins located at the surface of cancer cells. The latter mAbs are often combined with cytotoxic regimens, because they block survival of residual fractions of tumours that evade therapy-induced cell death. Antibodies, along with kinase inhibitors, have become in the last decade the mainstay of oncological pharmacology. However, partial and transient responses, as well as emergence of tumour resistance, currently limit clinical application of mAbs. To overcome these hurdles, oligoclonal antibody mixtures are being tested in animal models and in clinical trials. The first homo-combination of two mAbs, each engaging a distinct site of HER2, an oncogenic receptor tyrosine kinase (RTK), has been approved for treatment of breast cancer. Likewise, a hetero-combination of antibodies to two distinct T-cell antigens, PD1 and CTLA4, has been approved for treatment of melanoma. In a similar vein, additive or synergistic anti-tumour effects observed in animal models have prompted clinical testing of hetero-combinations of antibodies simultaneously engaging distinct RTKs. We discuss the promise of antibody cocktails reminiscent of currently used mixtures of chemotherapeutics and highlight mechanisms potentially underlying their enhanced clinical efficacy.
© 2016 The British Pharmacological Society.
Figures
Similar articles
-
Cancer Immunotherapy: The Dawn of Antibody Cocktails.Methods Mol Biol. 2019;1904:11-51. doi: 10.1007/978-1-4939-8958-4_2. Methods Mol Biol. 2019. PMID: 30539465 Review.
-
Promises and pitfalls for recombinant oligoclonal antibodies-based therapeutics in cancer and infectious disease.Curr Opin Immunol. 2016 Jun;40:51-61. doi: 10.1016/j.coi.2016.03.001. Epub 2016 Mar 23. Curr Opin Immunol. 2016. PMID: 26995095 Free PMC article. Review.
-
[Mimicking polyclonal immune response in therapy: from combination of two monoclonal antibodies to oligoclonal antibody-based mixtures].Med Sci (Paris). 2019 Dec;35(12):1083-1091. doi: 10.1051/medsci/2019216. Epub 2020 Jan 6. Med Sci (Paris). 2019. PMID: 31903921 Review. French.
-
Approaches to improve tumor accumulation and interactions between monoclonal antibodies and immune cells.MAbs. 2013 Jan-Feb;5(1):34-46. doi: 10.4161/mabs.22775. Epub 2012 Dec 4. MAbs. 2013. PMID: 23211740 Free PMC article. Review.
-
Future prospects of monoclonal antibodies as magic bullets in immunotherapy.Hum Antibodies. 2013;22(1-2):9-13. doi: 10.3233/HAB-130266. Hum Antibodies. 2013. PMID: 24284304 Review.
Cited by
-
Inhibitors of programmed cell death 1 (PD-1): a patent review (2010-2015).Expert Opin Ther Pat. 2016 Sep;26(9):973-7. doi: 10.1080/13543776.2016.1206527. Epub 2016 Jul 6. Expert Opin Ther Pat. 2016. PMID: 27367741 Free PMC article. No abstract available.
-
Discovery of BPR1K871, a quinazoline based, multi-kinase inhibitor for the treatment of AML and solid tumors: Rational design, synthesis, in vitro and in vivo evaluation.Oncotarget. 2016 Dec 27;7(52):86239-86256. doi: 10.18632/oncotarget.13369. Oncotarget. 2016. PMID: 27863392 Free PMC article.
-
The IUPHAR Guide to Immunopharmacology: connecting immunology and pharmacology.Immunology. 2020 May;160(1):10-23. doi: 10.1111/imm.13175. Epub 2020 Mar 2. Immunology. 2020. PMID: 32020584 Free PMC article. Review.
-
Aptamers and Glioblastoma: Their Potential Use for Imaging and Therapeutic Applications.Int J Mol Sci. 2017 Nov 30;18(12):2576. doi: 10.3390/ijms18122576. Int J Mol Sci. 2017. PMID: 29189740 Free PMC article. Review.
-
Combination of immune checkpoint blockade with DNA cancer vaccine induces potent antitumor immunity against P815 mastocytoma.Sci Rep. 2018 Oct 24;8(1):15732. doi: 10.1038/s41598-018-33933-7. Sci Rep. 2018. PMID: 30356111 Free PMC article.
References
-
- Abella JV, Park M (2009). Breakdown of endocytosis in the oncogenic activation of receptor tyrosine kinases. Am J Physiol Endocrinol Metab 296: E973–E984. - PubMed
-
- Aboud‐Pirak E, Hurwitz E, Pirak ME, Bellot F, Schlessinger J, Sela M (1988). Efficacy of antibodies to epidermal growth factor receptor against KB carcinoma in vitro and in nude mice. J Natl Cancer Inst 80: 1605–1611. - PubMed
-
- Amado RG, Wolf M, Peeters M, Van Cutsem E, Siena S, Freeman DJ, et al. (2008). Wild‐type KRAS is required for panitumumab efficacy in patients with metastatic colorectal cancer. J Clin Oncol 26: 1626–1634. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

