Multiple Domains of GlcNAc-1-phosphotransferase Mediate Recognition of Lysosomal Enzymes
- PMID: 26833567
- PMCID: PMC4825028
- DOI: 10.1074/jbc.M116.714568
Multiple Domains of GlcNAc-1-phosphotransferase Mediate Recognition of Lysosomal Enzymes
Abstract
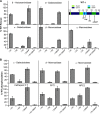
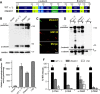
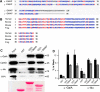
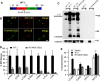
The Golgi enzyme UDP-GlcNAc:lysosomal enzymeN-acetylglucosamine-1-phosphotransferase (GlcNAc-1-phosphotransferase), an α2β2γ2hexamer, mediates the initial step in the addition of the mannose 6-phosphate targeting signal on newly synthesized lysosomal enzymes. This tag serves to direct the lysosomal enzymes to lysosomes. A key property of GlcNAc-1-phosphotransferase is its unique ability to distinguish the 60 or so lysosomal enzymes from the numerous non-lysosomal glycoproteins with identical Asn-linked glycans. In this study, we demonstrate that the two Notch repeat modules and the DNA methyltransferase-associated protein interaction domain of the α subunit are key components of this recognition process. Importantly, different combinations of these domains are involved in binding to individual lysosomal enzymes. This study also identifies the γ-binding site on the α subunit and demonstrates that in the majority of instances the mannose 6-phosphate receptor homology domain of the γ subunit is required for optimal phosphorylation. These findings serve to explain how GlcNAc-1-phosphotransferase recognizes a large number of proteins that lack a common structural motif.
Keywords: lysosomal glycoprotein; lysosomal storage disease; lysosome; mannose 6-phosphate (Man-6-P); phosphorylation.
© 2016 by The American Society for Biochemistry and Molecular Biology, Inc.
Figures

Similar articles
-
Analysis of mucolipidosis II/III GNPTAB missense mutations identifies domains of UDP-GlcNAc:lysosomal enzyme GlcNAc-1-phosphotransferase involved in catalytic function and lysosomal enzyme recognition.J Biol Chem. 2015 Jan 30;290(5):3045-56. doi: 10.1074/jbc.M114.612507. Epub 2014 Dec 11. J Biol Chem. 2015. PMID: 25505245 Free PMC article.
-
The DMAP interaction domain of UDP-GlcNAc:lysosomal enzyme N-acetylglucosamine-1-phosphotransferase is a substrate recognition module.Proc Natl Acad Sci U S A. 2013 Jun 18;110(25):10246-51. doi: 10.1073/pnas.1308453110. Epub 2013 Jun 3. Proc Natl Acad Sci U S A. 2013. PMID: 23733939 Free PMC article.
-
Structural requirements for efficient processing and activation of recombinant human UDP-N-acetylglucosamine:lysosomal-enzyme-N-acetylglucosamine-1-phosphotransferase.J Biol Chem. 2006 Apr 28;281(17):11761-8. doi: 10.1074/jbc.M513717200. Epub 2006 Feb 28. J Biol Chem. 2006. PMID: 16507578
-
Molecular analysis of the GlcNac-1-phosphotransferase.J Inherit Metab Dis. 2008 Apr;31(2):253-7. doi: 10.1007/s10545-008-0862-5. Epub 2008 Apr 15. J Inherit Metab Dis. 2008. PMID: 18425436 Review.
-
[Lysosomal hydrolases have specific conformational domains for acquisition of mannose-6-phosphate].Nihon Rinsho. 1995 Dec;53(12):2892-7. Nihon Rinsho. 1995. PMID: 8577031 Review. Japanese.
Cited by
-
Differential recognition of oligomannose isomers by glycan-binding proteins involved in innate and adaptive immunity.Sci Adv. 2021 Jun 9;7(24):eabf6834. doi: 10.1126/sciadv.abf6834. Print 2021 Jun. Sci Adv. 2021. PMID: 34108208 Free PMC article.
-
Replication of Coxiella burnetii in a Lysosome-Like Vacuole Does Not Require Lysosomal Hydrolases.Infect Immun. 2019 Oct 18;87(11):e00493-19. doi: 10.1128/IAI.00493-19. Print 2019 Nov. Infect Immun. 2019. PMID: 31405956 Free PMC article.
-
Cell-autonomous expression of the acid hydrolase galactocerebrosidase.Proc Natl Acad Sci U S A. 2020 Apr 21;117(16):9032-9041. doi: 10.1073/pnas.1917675117. Epub 2020 Apr 6. Proc Natl Acad Sci U S A. 2020. PMID: 32253319 Free PMC article.
-
In Vivo Delivery of Therapeutic Molecules by Transplantation of Genome-Edited Induced Pluripotent Stem Cells.Cell Transplant. 2023 Jan-Dec;32:9636897231173734. doi: 10.1177/09636897231173734. Cell Transplant. 2023. PMID: 37183961 Free PMC article.
-
Identification of predominant GNPTAB gene mutations in Eastern Chinese patients with mucolipidosis II/III and a prenatal diagnosis of mucolipidosis II.Acta Pharmacol Sin. 2019 Feb;40(2):279-287. doi: 10.1038/s41401-018-0023-9. Epub 2018 Jun 5. Acta Pharmacol Sin. 2019. PMID: 29872134 Free PMC article.
References
-
- Reitman M. L., and Kornfeld S. (1981) Lysosomal enzyme targeting. N-Acetylglucosaminylphosphotransferase selectively phosphorylates native lysosomal enzymes. J. Biol. Chem. 256, 11977–11980 - PubMed
-
- Varki A., and Kornfeld S. (1980) Identification of a rat liver α-N-acetylglucosaminyl phosphodiesterase capable of removing “blocking” α-N-acetylglucosamine residues from phosphorylated high mannose oligosaccharides of lysosomal enzymes. J. Biol. Chem. 255, 8398–8401 - PubMed
-
- Bao M., Booth J. L., Elmendorf B. J., and Canfield W. M. (1996) Bovine UDP-N-acetylglucosamine:lysosomal-enzyme N-acetylglucosamine-1-phosphotransferase. I. Purification and subunit structure. J. Biol. Chem. 271, 31437–31445 - PubMed
-
- Kudo M., Bao M., D'Souza A., Ying F., Pan H., Roe B. A., and Canfield W. M. (2005) The α- and β-subunits of the human UDP-N-acetylglucosamine:lysosomal enzyme N-acetylglucosamine-1-phosphotransferase [corrected] are encoded by a single cDNA. J. Biol. Chem. 280, 36141–36149 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

