Species variation in the degree of sex differences in brain and behaviour related to birdsong: adaptations and constraints
- PMID: 26833837
- PMCID: PMC4785902
- DOI: 10.1098/rstb.2015.0117
Species variation in the degree of sex differences in brain and behaviour related to birdsong: adaptations and constraints
Abstract
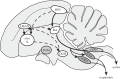
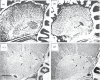
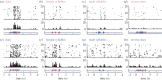
The song-control system, a neural circuit that controls the learning and production of birdsong, provided the first example in vertebrates of prominent macro-morphological sex differences in the brain. Forebrain nuclei HVC, robust nucleus of the arcopallium (RA) and area X all exhibit prominent male-biased sex differences in volume in zebra finches and canaries. Subsequent studies compared species that exhibited different degrees of a sex difference in song behaviour and revealed an overall positive correlation between male biases in song behaviour and male biases in the volume of the song nuclei. However, several exceptions have been described in which male biases in HVC and RA are observed even though song behaviour is equal or even female-biased. Other phenotypic measures exhibit lability in both sexes. In the duetting plain-tailed wren (Pheugopedius euophrys), males and females have auditory cells in the song system that are tuned to the joint song the two sexes produce rather than just male or female components. These findings suggest that there may be constraints on the adaptive response of the song system to ecological conditions as assessed by nucleus volume but that other critical variables regulating song can respond so that each sex can modify its song behaviour as needed.
Keywords: HVC; canary; sexual selection; song-control system.
© 2016 The Author(s).
Figures
Similar articles
-
Sex differences and similarities in the neural circuit regulating song and other reproductive behaviors in songbirds.Neurosci Biobehav Rev. 2020 Nov;118:258-269. doi: 10.1016/j.neubiorev.2020.07.026. Epub 2020 Jul 28. Neurosci Biobehav Rev. 2020. PMID: 32735803 Free PMC article. Review.
-
Sex differences in neuropeptide staining of song-control nuclei in zebra finch brains.Brain Behav Evol. 1997;50(5):284-303. doi: 10.1159/000113342. Brain Behav Evol. 1997. PMID: 9360005
-
Hormone accumulation in song regions of the canary brain.J Neurobiol. 1992 Sep;23(7):871-80. doi: 10.1002/neu.480230708. J Neurobiol. 1992. PMID: 1431849
-
Testosterone-induced changes in adult canary brain are reversible.J Neurobiol. 1993 May;24(5):627-40. doi: 10.1002/neu.480240508. J Neurobiol. 1993. PMID: 7686962
-
Sex differences in songbirds 25 years later: what have we learned and where do we go?Microsc Res Tech. 2001 Sep 15;54(6):327-34. doi: 10.1002/jemt.1146. Microsc Res Tech. 2001. PMID: 11668646 Review.
Cited by
-
Sex differences and similarities in the neural circuit regulating song and other reproductive behaviors in songbirds.Neurosci Biobehav Rev. 2020 Nov;118:258-269. doi: 10.1016/j.neubiorev.2020.07.026. Epub 2020 Jul 28. Neurosci Biobehav Rev. 2020. PMID: 32735803 Free PMC article. Review.
-
Neuroendocrine-Immune Crosstalk Shapes Sex-Specific Brain Development.Endocrinology. 2020 Jun 1;161(6):bqaa055. doi: 10.1210/endocr/bqaa055. Endocrinology. 2020. PMID: 32270188 Free PMC article. Review.
-
Neuroendocrine and behavioral response to testosterone-induced female song in canaries (Serinus canaria).Physiol Behav. 2022 Jun 1;250:113782. doi: 10.1016/j.physbeh.2022.113782. Epub 2022 Mar 18. Physiol Behav. 2022. PMID: 35314175 Free PMC article.
-
New insights from female bird song: towards an integrated approach to studying male and female communication roles.Biol Lett. 2019 Apr 26;15(4):20190059. doi: 10.1098/rsbl.2019.0059. Biol Lett. 2019. PMID: 30940020 Free PMC article.
-
Testosterone or Estradiol When Implanted in the Medial Preoptic Nucleus Trigger Short Low-Amplitude Songs in Female Canaries.eNeuro. 2019 May 8;6(2):ENEURO.0502-18.2019. doi: 10.1523/ENEURO.0502-18.2019. Print 2019 Mar/Apr. eNeuro. 2019. PMID: 31068363 Free PMC article.
References
-
- Nottebohm F. 1980. Brain pathways for vocal learning in birds: a review of the first 10 years. In Progress in psychobiology and physiological psychology (eds Sprague JM, Epstein AN), pp. 85–214. New York, NY: Academic Press.
-
- Nottebohm F. 2015. Fernando Nottebohm. In History of neuroscience in autobiography, vol. 8 (ed. Squire L), pp. 324–360. Washington, DC: Society for Neuroscience.
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources

