Minocycline-Induced Cutaneous Hyperpigmentation in an Orthopedic Patient Population
- PMID: 26835479
- PMCID: PMC4731832
- DOI: 10.1093/ofid/ofv107
Minocycline-Induced Cutaneous Hyperpigmentation in an Orthopedic Patient Population
Abstract
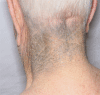
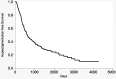
Background. The objectives of this study were to estimate the incidence and evaluate risk factors for development of minocycline-induced cutaneous hyperpigmentation in patients with orthopedic infections. Methods. Patients with orthopedic infections evaluated at Mayo Clinic (Rochester, MN) and treated with minocycline from 1 January 2002 to 31 December 2011 were retrospectively identified. Long-term minocycline suppression was defined as daily minocycline use for at least 3 months. A proportional hazards model was used to evaluate potential risk factors. Results. Of 291 patients receiving long-term minocycline suppression, 54% (156 of 291) developed hyperpigmentation after a mean follow-up of 4.8 years (range, 0.3-13.2 years); 88% involved blue-gray pigmentation of normal skin that appeared most commonly in the lower (75%) and upper extremities (44%). The mean duration of minocycline therapy before hyperpigmentation was 1.5 years (range, 0.1-9 years) with a mean cumulative dosage of 107.3 g (range, 8.6-657 g). Notable risk factors include a history of vitamin D deficiency (relative risk [RR], 6.29; 95% confidence interval [CI], 1.91-15.27; P = .0052), presence of a shoulder prosthesis (RR, 3.2; 95% CI, 1.23-6.56; P = .0062), noncirrhotic liver pathology (RR, 3.63; 95% CI, 1.11-8.75; P = .0359), and use of a concurrent medication also known to cause hyperpigmentation (RR, 4.75; 95% CI, 1.83-10.1; P = .0029). Conclusions. Hyperpigmentation associated with the use of long-term minocycline suppression in patients with orthopedic infections is common.
Keywords: antimicrobial side effects; minocycline; orthopedic infections.
Figures
Similar articles
-
Minocycline-induced hyperpigmentation in rheumatoid arthritis.J Clin Rheumatol. 2008 Feb;14(1):17-20. doi: 10.1097/RHU.0b013e31816356d1. J Clin Rheumatol. 2008. PMID: 18431092
-
Minocycline-induced hyperpigmentation in patients with pemphigus and pemphigoid.Arch Dermatol. 2000 Sep;136(9):1133-8. doi: 10.1001/archderm.136.9.1133. Arch Dermatol. 2000. PMID: 10987869
-
Minocycline-induced hyperpigmentation: comparison of 3 Q-switched lasers to reverse its effects.Clin Cosmet Investig Dermatol. 2013 May 31;6:159-62. doi: 10.2147/CCID.S42166. Print 2013. Clin Cosmet Investig Dermatol. 2013. PMID: 23754872 Free PMC article.
-
Minocycline-induced hyperpigmentation of the tongue: successful treatment with the Q-switched ruby laser.Dermatol Surg. 2002 Mar;28(3):205-9. doi: 10.1046/j.1524-4725.2002.01083.x. Dermatol Surg. 2002. PMID: 11896769 Review.
-
Extensive cutaneous hyperpigmentation caused by minocycline.J Am Acad Dermatol. 1993 Feb;28(2 Pt 2):292-5. doi: 10.1016/0190-9622(93)70037-t. J Am Acad Dermatol. 1993. PMID: 8436641 Review.
Cited by
-
Minocycline-induced skin pigmentation.Infection. 2017 Feb;45(1):129-130. doi: 10.1007/s15010-016-0952-5. Epub 2016 Oct 17. Infection. 2017. PMID: 27753000 No abstract available.
-
A Severe Case of Minocycline-induced Hyperpigmentation of the Lower Extremities.Cureus. 2018 May 22;10(5):e2672. doi: 10.7759/cureus.2672. Cureus. 2018. PMID: 30050728 Free PMC article.
-
Minocycline-Induced Hyperpigmentation: Importance of Early Diagnosis.IDCases. 2023 Oct 5;34:e01901. doi: 10.1016/j.idcr.2023.e01901. eCollection 2023. IDCases. 2023. PMID: 37841948 Free PMC article.
-
Diffuse skin hyperpigmentation associated with chronic minocycline use in a patient with prosthetic joint infection.IDCases. 2016 Dec 19;7:30-31. doi: 10.1016/j.idcr.2016.12.003. eCollection 2017. IDCases. 2016. PMID: 28050350 Free PMC article.
-
Minocycline-Induced Gum Pigmentation during Treatment for Acne Vulgaris.Case Rep Pediatr. 2022 Oct 14;2022:9493061. doi: 10.1155/2022/9493061. eCollection 2022. Case Rep Pediatr. 2022. PMID: 36276924 Free PMC article.
References
-
- Patterson JW, Wilson B, Wick MR, Heath C. Hyperpigmented scar due to minocycline therapy. Cutis 2004; 74:293–8. - PubMed
-
- Bowen AR, McCalmont TH. The histopathology of subcutaneous minocycline pigmentation. J Am Acad Dermatol 2007; 57:836–9. - PubMed
-
- Dwyer CM, Cuddihy AM, Kerr RE et al. . Skin pigmentation due to minocycline treatment of facial dermatoses. Br J Dermatol 1993; 129:158–62. - PubMed
-
- Goulden V, Glass D, Cunliffe WJ. Safety of long-term high-dose minocycline in the treatment of acne. Br J Dermatol 1996; 134:693–5. - PubMed
-
- Roberts G, Capell HA. The frequency and distribution of minocycline induced hyperpigmentation in a rheumatoid arthritis population. J Rheumatol 2006; 33:1254–7. - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources

