Bioadsorption of Rare Earth Elements through Cell Surface Display of Lanthanide Binding Tags
- PMID: 26836847
- PMCID: PMC5381720
- DOI: 10.1021/acs.est.5b06129
Bioadsorption of Rare Earth Elements through Cell Surface Display of Lanthanide Binding Tags
Abstract
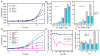
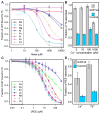
With the increasing demand for rare earth elements (REEs) in many emerging clean energy technologies, there is an urgent need for the development of new approaches for efficient REE extraction and recovery. As a step toward this goal, we genetically engineered the aerobic bacterium Caulobacter crescentus for REE adsorption through high-density cell surface display of lanthanide binding tags (LBTs) on its S-layer. The LBT-displayed strains exhibited enhanced adsorption of REEs compared to cells lacking LBT, high specificity for REEs, and an adsorption preference for REEs with small atomic radii. Adsorbed Tb(3+) could be effectively recovered using citrate, consistent with thermodynamic speciation calculations that predicted strong complexation of Tb(3+) by citrate. No reduction in Tb(3+) adsorption capacity was observed following citrate elution, enabling consecutive adsorption/desorption cycles. The LBT-displayed strain was effective for extracting REEs from the acid leachate of core samples collected at a prospective rare earth mine. Our collective results demonstrate a rapid, efficient, and reversible process for REE adsorption with potential industrial application for REE enrichment and separation.
Figures


Similar articles
-
Effectively auto-regulated adsorption and recovery of rare earth elements via an engineered E. coli.J Hazard Mater. 2022 Feb 15;424(Pt C):127642. doi: 10.1016/j.jhazmat.2021.127642. Epub 2021 Oct 30. J Hazard Mater. 2022. PMID: 34775317
-
Recovery of Rare Earth Elements from Low-Grade Feedstock Leachates Using Engineered Bacteria.Environ Sci Technol. 2017 Nov 21;51(22):13471-13480. doi: 10.1021/acs.est.7b02414. Epub 2017 Nov 2. Environ Sci Technol. 2017. PMID: 28944666
-
Microbe Encapsulation for Selective Rare-Earth Recovery from Electronic Waste Leachates.Environ Sci Technol. 2019 Dec 3;53(23):13888-13897. doi: 10.1021/acs.est.9b04608. Epub 2019 Nov 21. Environ Sci Technol. 2019. PMID: 31702144
-
Gravity-Driven Separation for Enrichment of Rare Earth Elements Using Lanthanide Binding Peptide-Immobilized Resin.ACS Appl Bio Mater. 2024 Dec 16;7(12):7828-7837. doi: 10.1021/acsabm.3c01280. Epub 2024 Apr 29. ACS Appl Bio Mater. 2024. PMID: 38685483 Review.
-
Global demand for rare earth resources and strategies for green mining.Environ Res. 2016 Oct;150:182-190. doi: 10.1016/j.envres.2016.05.052. Epub 2016 Jun 10. Environ Res. 2016. PMID: 27295408 Review.
Cited by
-
Genomic characterization of rare earth binding by Shewanella oneidensis.Sci Rep. 2023 Sep 25;13(1):15975. doi: 10.1038/s41598-023-42742-6. Sci Rep. 2023. PMID: 37749198 Free PMC article.
-
Enhanced rare-earth separation with a metal-sensitive lanmodulin dimer.Nature. 2023 Jun;618(7963):87-93. doi: 10.1038/s41586-023-05945-5. Epub 2023 May 31. Nature. 2023. PMID: 37259003 Free PMC article.
-
Recovery of rare earth elements from low-grade coal fly ash using a recyclable protein biosorbent.Front Bioeng Biotechnol. 2024 May 16;12:1385845. doi: 10.3389/fbioe.2024.1385845. eCollection 2024. Front Bioeng Biotechnol. 2024. PMID: 38817924 Free PMC article.
-
The Role of Asparagine as a Gatekeeper Residue in the Selective Binding of Rare Earth Elements by Lanthanide-Binding Peptides.Chemistry. 2025 Jun 12;31(33):e202501318. doi: 10.1002/chem.202501318. Epub 2025 May 9. Chemistry. 2025. PMID: 40312258 Free PMC article.
-
Emerging enzyme surface display systems for waste resource recovery.Environ Microbiol. 2023 Feb;25(2):241-249. doi: 10.1111/1462-2920.16284. Epub 2022 Nov 21. Environ Microbiol. 2023. PMID: 36369958 Free PMC article. Review.
References
-
- Hennebel T, Boon N, Maes S, Lenz M. Biotechnologies for critical raw material recovery from primary and secondary sources: R&D priorities and future perspectives. New biotechnology. 2015;32(1):121–7. - PubMed
-
- DOE. Critical Materials Strategy. 2011 http://www.energy.gov.
-
- Xie F, Zhang TA, Dreisinger D, Doyle F. A critical review on solvent extraction of rare earths from aqueous solutions. Miner Eng. 2014;56:10–28.
-
- Nash KL, Jensen MP. Analytical-scale separations of the lanthanides: A review of techniques and fundamentals. Separ Sci Technol. 2001;36(5–6):1257–1282.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

