Protease-activated Receptor-2 (PAR-2)-mediated Nf-κB Activation Suppresses Inflammation-associated Tumor Suppressor MicroRNAs in Oral Squamous Cell Carcinoma
- PMID: 26839311
- PMCID: PMC4807278
- DOI: 10.1074/jbc.M115.692640
Protease-activated Receptor-2 (PAR-2)-mediated Nf-κB Activation Suppresses Inflammation-associated Tumor Suppressor MicroRNAs in Oral Squamous Cell Carcinoma
Abstract
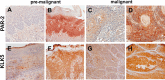
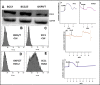
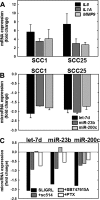
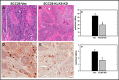
Oral cancer is the sixth most common cause of death from cancer with an estimated 400,000 deaths worldwide and a low (50%) 5-year survival rate. The most common form of oral cancer is oral squamous cell carcinoma (OSCC). OSCC is highly inflammatory and invasive, and the degree of inflammation correlates with tumor aggressiveness. The G protein-coupled receptor protease-activated receptor-2 (PAR-2) plays a key role in inflammation. PAR-2 is activated via proteolytic cleavage by trypsin-like serine proteases, including kallikrein-5 (KLK5), or by treatment with activating peptides. PAR-2 activation induces G protein-α-mediated signaling, mobilizing intracellular calcium and Nf-κB signaling, leading to the increased expression of pro-inflammatory mRNAs. Little is known, however, about PAR-2 regulation of inflammation-related microRNAs. Here, we assess PAR-2 expression and function in OSCC cell lines and tissues. Stimulation of PAR-2 activates Nf-κB signaling, resulting in RelA nuclear translocation and enhanced expression of pro-inflammatory mRNAs. Concomitantly, suppression of the anti-inflammatory tumor suppressor microRNAs let-7d, miR-23b, and miR-200c was observed following PAR-2 stimulation. Analysis of orthotopic oral tumors generated by cells with reduced KLK5 expression showed smaller, less aggressive lesions with reduced inflammatory infiltrate relative to tumors generated by KLK5-expressing control cells. Together, these data support a model wherein KLK5-mediated PAR-2 activation regulates the expression of inflammation-associated mRNAs and microRNAs, thereby modulating progression of oral tumors.
Keywords: NF-κB; inflammation; kallikrein; microRNA (miRNA); oral cancer; protease; protease-activated receptor; serine protease.
© 2016 by The American Society for Biochemistry and Molecular Biology, Inc.
Figures


References
-
- World Health Organization (2012) GLOBOCAN 2012: Estimated cancer incidence, mortality and prevalence worldwide in 2012 (globocan.iarc.fr/Pages/fact_sheets_cancer.aspx)
-
- Neville B. W., and Day T. A. (2002) Oral cancer and precancerous lesions. CA Cancer J. Clin. 52, 195–215 - PubMed
-
- Kramer I. R., Lucas R. B., Pindborg J. J., and Sobin L. H. (1978) Definition of leukoplakia and related lesions: an aid to studies on oral precancer. Oral Surg. Oral Med. Oral Pathol. 46, 518–539 - PubMed
-
- Lumerman H., Freedman P., and Kerpel S. (1995) Oral epithelial dysplasia and the development of invasive squamous cell carcinoma. Oral Surg. Oral Med. Oral Pathol. Oral Radiol Endod. 79, 321–329 - PubMed
-
- Vedtofte P., Holmstrup P., Hjorting-Hansen E., and Pindborg J. J. (1987) Surgical treatment of premalignant lesions of the oral mucosa. Int. J. Oral Maxillofac. Surg. 16, 656–664 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

