Carbon Nanotube and Asbestos Exposures Induce Overlapping but Distinct Profiles of Lung Pathology in Non-Swiss Albino CF-1 Mice
- PMID: 26839332
- PMCID: PMC4976500
- DOI: 10.1177/0192623315620587
Carbon Nanotube and Asbestos Exposures Induce Overlapping but Distinct Profiles of Lung Pathology in Non-Swiss Albino CF-1 Mice
Abstract
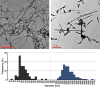
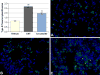
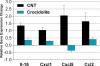
Carbon nanotubes (CNTs) are emerging as important occupational and environmental toxicants owing to their increasing prevalence and potential to be inhaled as airborne particles. CNTs are a concern because of their similarities to asbestos, which include fibrous morphology, high aspect ratio, and biopersistence. Limitations in research models have made it difficult to experimentally ascertain the risk of CNT exposures to humans and whether these may lead to lung diseases classically associated with asbestos, such as mesothelioma and fibrosis. In this study, we sought to comprehensively compare profiles of lung pathology in mice following repeated exposures to multiwall CNTs or crocidolite asbestos (CA). We show that both exposures resulted in granulomatous inflammation and increased interstitial collagen; CA exposures caused predominantly bronchoalveolar hyperplasia, whereas CNT exposures caused alveolar hyperplasia of type II pneumocytes (T2Ps). T2Ps isolated from CNT-exposed lungs were found to have upregulated proinflammatory genes, including interleukin 1ß (IL-1ß), in contrast to those from CA exposed. Immunostaining in tissue showed that while both toxicants increased IL-1ß protein expression in lung cells, T2P-specific IL-1ß increases were greater following CNT exposure. These results suggest related but distinct mechanisms of action by CNTs versus asbestos which may lead to different outcomes in the 2 exposure types.
Keywords: carbon nanotubes; comparative pathology; crocidolite asbestos; fiber toxicology; lung; mouse.
© The Author(s) 2016.
Figures







References
-
- Adamson IY, Bowden DH. Response of mouse lung to crocidolite asbestos. 2. Pulmonary fibrosis after long fibres. The Journal of pathology. 1987;152(2):109–117. - PubMed
-
- Akiyama I, Ogami A, Oyabu T, Yamato H, Morimoto Y, Tanaka I. Pulmonary effects and biopersistence of deposited silicon carbide whisker after 1-year inhalation in rats. Inhalation toxicology. 2007;19(2):141–147. - PubMed
-
- Birch ME. Monitoring of diesel particulate exhaust in the workplace. In: Schlecht PC, O’Connor PF, editors. NIOSH Manual of Analytical Methods (NMAM) 4th. Department of Health and Human Services, Public Health Service, Center for Disease Control and Prevention, National Institute for Occupational Safety and Health. DHHS(NIOSH); 154, Cincinnati, Ohio: 2004a. Third Supplement to NMAM.
-
- Birch ME. NIOSH Method 5040 update. In: Schlecht PC, O’Connor PF, editors. NIOSH Manual of Analytical Methods (NMAM) 4th. Department of Health and Human Services, Public Health Service, Center for Disease Control and Prevention, National Institute for Occupational Safety and Health. DHHS(NIOSH); 154, Cincinnati, Ohio: 2004b. Third Supplement to NMAM.
-
- Bonner JC, Silva RM, Taylor AJ, Brown JM, Hilderbrand SC, Castranova V, Porter D, Elder A, Oberdörster G, Harkema JR, Bramble LA, Kavanagh TJ, Botta D, Nei A, Pinkerton KE. Interlaboratory evaluation of rodent pulmonary responses to engineered nanomaterials: the NIEHS Nano GO Consortium. Environmental health perspectives. 2013;121(6):676–682. - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

