Cost-Effective Isolation of a Process Impurity of Pregabalin
- PMID: 26839830
- PMCID: PMC4727790
- DOI: 10.3797/scipharm.1501-16
Cost-Effective Isolation of a Process Impurity of Pregabalin
Abstract
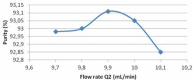
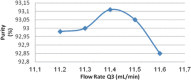
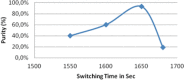
Cost-effective isolation methods were developed on preparative HPLC, flash LC, and simulated moving bed (SMB) to prepare the process impurity, 3-(aminomethyl)-5-methylhex-4-enoic acid (4-ene impurity), of pregabalin. By a thorough experimental study on the different isolation techniques available, it was concluded that SMB was the most cost-effective. Hence, it was a continuous chromatography that utilized the advantage of SMB so that a high quantity of the impurity was generated in a short period of time. SMB was equipped with eight reversed-phased columns and was used to separate the process impurity of pregabalin. The effects of flow rate in zone 2 (Q2) and 3 (Q3), as well as switching time, on the operating performance parameters like purity, productivity, and desorbent consumption were studied. Operating conditions leading to more than 90% purity in the raffinate outlet stream were identified, together with those achieving optimal performance. All of these developed methods are novel, cost-effective, and can be applied to the isolation of other process- and stability-related impurities of pregabalin.
Keywords: Flash chromatography; Impurity; Isolation; Pregabalin; Preparative HPLC; Simulating moving bed.
Figures
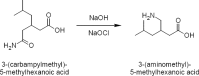
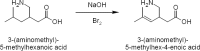

Similar articles
-
Three-port operation in three-zone simulated moving bed chromatography.J Chromatogr A. 2014 May 2;1340:79-89. doi: 10.1016/j.chroma.2014.03.012. Epub 2014 Mar 11. J Chromatogr A. 2014. PMID: 24661870
-
Separation of phosphatidylcholine from soybean phospholipids by simulated moving bed.J Zhejiang Univ Sci B. 2006 Jul;7(7):559-64. doi: 10.1631/jzus.2006.B0559. J Zhejiang Univ Sci B. 2006. PMID: 16773730 Free PMC article.
-
Partial port-closing strategy for obtaining high throughput or high purities in a four-zone simulated moving bed chromatography for binary separation.J Chromatogr A. 2010 Oct 15;1217(42):6522-30. doi: 10.1016/j.chroma.2010.08.049. Epub 2010 Aug 26. J Chromatogr A. 2010. PMID: 20837353
-
Simulated moving bed chromatography for the separation of enantiomers.J Chromatogr A. 2009 Jan 23;1216(4):709-38. doi: 10.1016/j.chroma.2008.10.075. Epub 2008 Nov 1. J Chromatogr A. 2009. PMID: 19004446 Review.
-
Impurity Profile of Bronchodilators used in Asthma: A Critical Review.Curr Drug Discov Technol. 2018;15(4):272-304. doi: 10.2174/1570163814666170829141614. Curr Drug Discov Technol. 2018. PMID: 28875855 Review.
References
-
- WebMD. Pregabalin. http://www.webmd.com/drugs/drug-93954-pregabalin+oral.aspx .
-
- Moore RA, Straube S, Wiffen PJ, Derry S, McQuay Pregabalin for acute and chronic pain in adults. Cochrane Database Syst Rev. 2009. p. CD007076. http://dx.doi.org/10.1002/14651858.CD007076.pub2 . - PMC - PubMed
-
- Wensel TM, Powe KW, Cates ME. Pregabalin for the treatment of generalized anxiety disorder. Ann Pharmacother. 2012;46:424–429. http://dx.doi.org/10.1345/aph.1Q405 . - PubMed
-
- Bandelow B, Wedekind D, Leon T. Pregabalin for the treatment of generalized anxiety disorder: a novel pharmacologic intervention. Expert Rev Neurother. 2007;7:769–781. http://dx.doi.org/10.1586/14737175.7.7.769 . - PubMed
-
- Owen RT. Pregabalin: its efficacy, safety and tolerability profile in generalized anxiety. Drugs Today. 2007;43:601–610. http://dx.doi.org/10.1358/dot.2007.43.9.1133188 . - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
