To What Extent is FAIMS Beneficial in the Analysis of Proteins?
- PMID: 26843211
- PMCID: PMC4792363
- DOI: 10.1007/s13361-015-1326-4
To What Extent is FAIMS Beneficial in the Analysis of Proteins?
Abstract
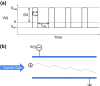
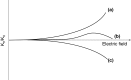
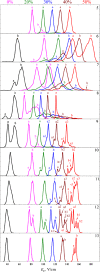
High field asymmetric waveform ion mobility spectrometry (FAIMS), also known as differential ion mobility spectrometry, is emerging as a tool for biomolecular analysis. In this article, the benefits and limitations of FAIMS for protein analysis are discussed. The principles and mechanisms of FAIMS separation of ions are described, and the differences between FAIMS and conventional ion mobility spectrometry are detailed. Protein analysis is considered from both the top-down (intact proteins) and the bottom-up (proteolytic peptides) perspective. The roles of FAIMS in the analysis of complex mixtures of multiple intact proteins and in the analysis of multiple conformers of a single protein are assessed. Similarly, the application of FAIMS in proteomics and targeted analysis of peptides are considered.
Keywords: DMS; FAIMS; Peptides; Proteins; Proteomics.
Figures

Similar articles
-
High-field asymmetric waveform ion mobility spectrometry for mass spectrometry-based proteomics.Expert Rev Proteomics. 2012 Oct;9(5):505-17. doi: 10.1586/epr.12.50. Expert Rev Proteomics. 2012. PMID: 23194268 Free PMC article. Review.
-
Distortion of ion structures by field asymmetric waveform ion mobility spectrometry.Anal Chem. 2007 Feb 15;79(4):1523-8. doi: 10.1021/ac061306c. Anal Chem. 2007. PMID: 17297950
-
Enhancement of mass spectrometry performance for proteomic analyses using high-field asymmetric waveform ion mobility spectrometry (FAIMS).J Mass Spectrom. 2015 Nov;50(11):1181-95. doi: 10.1002/jms.3646. J Mass Spectrom. 2015. PMID: 26505763
-
High-field asymmetric waveform ion mobility spectrometry: a new tool for mass spectrometry.J Chromatogr A. 2004 Nov 26;1058(1-2):3-19. J Chromatogr A. 2004. PMID: 15595648 Review.
-
Elimination of the helium requirement in high-field asymmetric waveform ion mobility spectrometry (FAIMS): beneficial effects of decreasing the analyzer gap width on peptide analysis.Rapid Commun Mass Spectrom. 2011 Jul 30;25(14):1959-71. doi: 10.1002/rcm.5078. Rapid Commun Mass Spectrom. 2011. PMID: 21698679
Cited by
-
Identification of Plasmodium falciparum proteoforms from liver stage models.Malar J. 2020 Jan 7;19(1):10. doi: 10.1186/s12936-019-3093-3. Malar J. 2020. PMID: 31910830 Free PMC article.
-
Deeper Protein Identification Using Field Asymmetric Ion Mobility Spectrometry in Top-Down Proteomics.Anal Chem. 2021 Apr 27;93(16):6323-6328. doi: 10.1021/acs.analchem.1c00402. Epub 2021 Apr 12. Anal Chem. 2021. PMID: 33844503 Free PMC article.
-
Direct measurement of light and heavy antibody chains using ion mobility and middle-down mass spectrometry.MAbs. 2019 Nov-Dec;11(8):1351-1357. doi: 10.1080/19420862.2019.1668226. Epub 2019 Oct 13. MAbs. 2019. PMID: 31607219 Free PMC article.
-
Characterization of Complete Histone Tail Proteoforms Using Differential Ion Mobility Spectrometry.Anal Chem. 2017 May 16;89(10):5461-5466. doi: 10.1021/acs.analchem.7b00379. Epub 2017 Apr 26. Anal Chem. 2017. PMID: 28406606 Free PMC article.
-
Instrumentation at the Leading Edge of Proteomics.Anal Chem. 2024 May 21;96(20):7976-8010. doi: 10.1021/acs.analchem.3c04497. Epub 2024 May 13. Anal Chem. 2024. PMID: 38738990 Free PMC article. Review.
References
-
- Mason EA, McDaniel EW. Transport properties of ions in gases. New York: Wiley; 1988.
-
- Buryakov IA, Krylov EV, Nazarov EG, Rasulev UK. A new method of separation of multi-atomic ions by mobility at atmospheric pressure using a high-frequency amplitude-asymmetric strong electric field. Int. J. Mass Spectrom. Ion Processes. 1993;128:143–148. doi: 10.1016/0168-1176(93)87062-W. - DOI
-
- Purves RW, Guevremont R, Day S, Pipich CW, Matyjaszczyk M. Mass spectrometric chracterization of a high-field asymmetric waveform ion mobility spectrometer. Rev. Sci. Instrum. 1998;69:4094–4105. doi: 10.1063/1.1149255. - DOI
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

