Does the GH/IGF-1 axis contribute to skeletal sexual dimorphism? Evidence from mouse studies
- PMID: 26843472
- PMCID: PMC5488285
- DOI: 10.1016/j.ghir.2015.12.004
Does the GH/IGF-1 axis contribute to skeletal sexual dimorphism? Evidence from mouse studies
Abstract
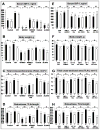
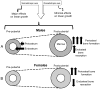
The contribution of the gonadotropic axis to skeletal sexual dimorphism (SSD) was clarified in recent years. Studies with animal models of estrogen receptor (ER) or androgen receptor (AR) null mice, as well as mice with bone cell-specific ablation of ER or AR, revealed that both hormones play major roles in skeletal acquisition, and that estrogen regulates skeletal accrual in both sexes. The growth hormone (GH) and its downstream effector, the insulin-like growth factor-1 (IGF-1) are also major determinants of peak bone mass during puberty and young adulthood, and play important roles in maintaining bone integrity during aging. A few studies in both humans and animal models suggest that in addition to the differences in sex steroid actions on bone, sex-specific effects of GH and IGF-1 play essential roles in SSD. However, the contributions of the somatotropic (GH/IGF-1) axis to SSD are controversial and data is difficult to interpret. GH/IGF-1 are pleotropic hormones that act in an endocrine and autocrine/paracrine fashion on multiple tissues, affecting body composition as well as metabolism. Thus, understanding the contribution of the somatotropic axis to SSD requires the use of mouse models that will differentiate between these two modes of action. Elucidation of the relative contribution of GH/IGF-1 axis to SSD is significant because GH is approved for the treatment of normal children with short stature and children with congenital growth disorders. Thus, if the GH/IGF-1 axis determines SSD, treatment with GH may be tailored according to sex. In the following review, we give an overview of the roles of sex steroids in determining SSD and how they may interact with the GH/IGF-1 axis in bone. We summarize several mouse models with impaired somatotropic axis and speculate on the possible contribution of that axis to SSD.
Keywords: Bone; Growth hormone receptor (GHR); Insulin-like growth factor-1 (IGF-1); Micro-computed tomography; Osteocyte; Parathyroid hormone (PTH); Skeletal sexual dimorphism (SSD).
Copyright © 2015 Elsevier Ltd. All rights reserved.
Figures

Similar articles
-
Regulation of skeletal growth and mineral acquisition by the GH/IGF-1 axis: Lessons from mouse models.Growth Horm IGF Res. 2016 Jun;28:26-42. doi: 10.1016/j.ghir.2015.09.004. Epub 2015 Sep 28. Growth Horm IGF Res. 2016. PMID: 26432542 Free PMC article. Review.
-
The role of GH/IGF-I-mediated mechanisms in sex differences in cortical bone size in mice.Calcif Tissue Int. 2011 Jan;88(1):1-8. doi: 10.1007/s00223-010-9436-2. Epub 2010 Nov 27. Calcif Tissue Int. 2011. PMID: 21113585 Free PMC article. Review.
-
Sex steroids, growth hormone, insulin-like growth factor-1: neuroendocrine and metabolic regulation in puberty.Horm Res. 1996;45(1-2):74-80. doi: 10.1159/000184763. Horm Res. 1996. PMID: 8742123 Review.
-
The anabolic effects of GH/IGF system on bone.Curr Pharm Des. 2004;10(21):2577-92. doi: 10.2174/1381612043383764. Curr Pharm Des. 2004. PMID: 15320746 Review.
-
Sexual dimorphism in cortical bone size and strength but not density is determined by independent and time-specific actions of sex steroids and IGF-1: evidence from pubertal mouse models.J Bone Miner Res. 2010 Mar;25(3):617-26. doi: 10.1359/jbmr.090828. J Bone Miner Res. 2010. PMID: 19888832
Cited by
-
The Somatotropic Axis in Human Aging: Framework for the Current State of Knowledge and Future Research.Cell Metab. 2016 Jun 14;23(6):980-989. doi: 10.1016/j.cmet.2016.05.014. Cell Metab. 2016. PMID: 27304500 Free PMC article. Review.
-
The Association Between IGF-I and IGFBP-3 and Incident Diabetes in an Older Population of Men and Women in the Cardiovascular Health Study.J Clin Endocrinol Metab. 2017 Dec 1;102(12):4541-4547. doi: 10.1210/jc.2017-01273. J Clin Endocrinol Metab. 2017. PMID: 29040592 Free PMC article.
-
Supplementation with >Your< Iron Syrup Corrects Iron Status in a Mouse Model of Diet-Induced Iron Deficiency.Biology (Basel). 2021 Apr 22;10(5):357. doi: 10.3390/biology10050357. Biology (Basel). 2021. PMID: 33922324 Free PMC article.
-
Endochondral Ossification for Spinal Fusion: A Novel Perspective from Biological Mechanisms to Clinical Applications.J Pers Med. 2024 Sep 9;14(9):957. doi: 10.3390/jpm14090957. J Pers Med. 2024. PMID: 39338212 Free PMC article. Review.
-
Long-Term Cola Intake Does Not Cause Evident Pathological Alterations in the Femoral Bone Microstructure: An Animal Study in Adult Mice.Nutrients. 2023 Jan 22;15(3):583. doi: 10.3390/nu15030583. Nutrients. 2023. PMID: 36771291 Free PMC article.
References
-
- Tanner JM, Whitehouse RH, Marubini E, Resele LF. The adolescent growth spurt of boys and girls of the Harpenden growth study. Ann Hum Biol. 1976;3:109–126. - PubMed
-
- Veldhuis JD, Roemmich JN, Richmond EJ, Rogol AD, Lovejoy JC, Sheffield-Moore M, Mauras N, Bowers CY. Endocrine control of body composition in infancy, childhood, and puberty. Endocr Rev. 2005;26:114–146. - PubMed
-
- Seeman E. Pathogenesis of bone fragility in women and men. Lancet. 2002;359:1841–1850. - PubMed
-
- Seeman E. Clinical review 137: Sexual dimorphism in skeletal size, density, and strength. J Clin Endocrinol Metab. 2001;86:4576–4584. - PubMed
-
- Schoenau E, Neu CM, Rauch F, Manz F. The development of bone strength at the proximal radius during childhood and adolescence. J Clin Endocrinol Metab. 2001;86:613–618. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

