Evaluating Shared Laboratory Services: Detecting Mycobacterium Tuberculosis Complex and Drug Resistance Using Molecular and Culture-Based Methods
- PMID: 26843677
- PMCID: PMC4716479
- DOI: 10.1177/003335491613100118
Evaluating Shared Laboratory Services: Detecting Mycobacterium Tuberculosis Complex and Drug Resistance Using Molecular and Culture-Based Methods
Abstract
Objectives: We explored sharing nucleic acid amplification testing (NAAT) for detection of Mycobacterium tuberculosis complex (MTBC) and molecular and phenotypic drug susceptibility testing between two state public health tuberculosis (TB) laboratories, and evaluated turnaround times and cost-effectiveness.
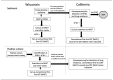
Methods: From September 1, 2012, to May 30, 2013, the Wisconsin State Laboratory of Hygiene (Wisconsin Lab) submitted specimens to the Microbial Diseases Laboratory of the California Department of Public Health (California Lab) for NAAT and molecular drug susceptibility testing (MDST) by pyrosequencing, and culture-based TB drug susceptibility testing by the BACTEC(TM) MGIT(TM) 960 system.
Results: A total of 182 specimens were referred to the California Lab, and 47 TB cases and 12 drug-resistant cases were identified. The average time for specimen transport was two days, which included one day for processing and packaging in the submitting laboratory. The average turnaround time for NAAT was 0.3 days at the Wisconsin Lab and 3.8 days at the California Lab, including time for specimen transport. Turnaround time for culture-based drug susceptibility testing increased by a median of 16 days when specimens were sent to the California Lab, but MDST results were reported in fewer than four days, a median of 22 days sooner than any culture-based drug susceptibility testing results.
Conclusion: This study revealed advantages and disadvantages associated with sharing services, and identified opportunities for improvement. Referral of specimens resulted in longer turnaround times for mycobacteriology test results and additional costs for transporting specimens. However, specialized testing such as pyrosequencing may not be available in low TB incidence areas, and these rapid results can have positive effects on patient management and TB control.
Figures
Similar articles
-
The Use of a Shared Services Model for Mycobacteriology Testing: Lessons Learned.Public Health Rep. 2018 Jan/Feb;133(1):93-99. doi: 10.1177/0033354917743498. Epub 2017 Dec 19. Public Health Rep. 2018. PMID: 29258383 Free PMC article.
-
[Mycobacterial tests].Kekkaku. 2008 Jan;83(1):43-59. Kekkaku. 2008. PMID: 18283915 Japanese.
-
A Model of Shared Mycobacteriology Testing Services: Lessons Learned.Public Health Rep. 2015 Nov-Dec;130(6):623-31. doi: 10.1177/003335491513000612. Public Health Rep. 2015. PMID: 26556934 Free PMC article.
-
Molecular genetic methods for diagnosis and antibiotic resistance detection of mycobacteria from clinical specimens.APMIS. 2004 Nov-Dec;112(11-12):728-52. doi: 10.1111/j.1600-0463.2004.apm11211-1203.x. APMIS. 2004. PMID: 15638836 Review.
-
Diagnostic mycobacteriology laboratory practices.Clin Infect Dis. 1995 Aug;21(2):291-9. doi: 10.1093/clinids/21.2.291. Clin Infect Dis. 1995. PMID: 8562734 Review.
Cited by
-
[Visualized detection for mycobacterium tuberculosis using loop-mediated isothermal amplification assay].Sheng Wu Yi Xue Gong Cheng Xue Za Zhi. 2017 Feb;34(1):72-7. doi: 10.7507/1001-5515.201606066. Sheng Wu Yi Xue Gong Cheng Xue Za Zhi. 2017. PMID: 29717591 Free PMC article. Chinese.
-
The Use of a Shared Services Model for Mycobacteriology Testing: Lessons Learned.Public Health Rep. 2018 Jan/Feb;133(1):93-99. doi: 10.1177/0033354917743498. Epub 2017 Dec 19. Public Health Rep. 2018. PMID: 29258383 Free PMC article.
-
Cost minimization analysis of line probe assay for detection of multidrug-resistant tuberculosis in Arkhangelsk region of Russian Federation.PLoS One. 2019 Jan 29;14(1):e0211203. doi: 10.1371/journal.pone.0211203. eCollection 2019. PLoS One. 2019. PMID: 30695043 Free PMC article.
-
Evaluation of the postal service for referral of specimen of drug resistance tuberculosis in Amhara region, Ethiopia; mixed method.Afr Health Sci. 2021 Jun;21(2):619-627. doi: 10.4314/ahs.v21i2.17. Afr Health Sci. 2021. PMID: 34795715 Free PMC article.
-
Treatment outcomes of children and adolescents receiving drug-resistant TB treatment in a routine TB programme, Mumbai, India.PLoS One. 2021 Feb 18;16(2):e0246639. doi: 10.1371/journal.pone.0246639. eCollection 2021. PLoS One. 2021. PMID: 33600431 Free PMC article.
References
-
- World Health Organization. Geneva: WHO; 2014. Global tuberculosis report 2013. Also available from: http://apps.who.int/iris/bitstream/10665/91355/1/9789241564656_eng.pdf [cited 2015 Jan 30]
-
- Centers for Disease Control and Prevention (US) Atlanta: CDC; 2014. Reported tuberculosis in the United States, 2013. Also available from: http://www.cdc.gov/features/dstuberculosis [cited 2015 Sep 3]
-
- Kent PT, Kubica GP. Atlanta: Centers for Disease Control and Prevention (US); 1985. Public health mycobacteriology: a guide for the level III laboratory.
-
- Halse TA, Edwards J, Cunningham PL, Wolfgang WJ, Dumas NB, Escuyer VE, et al. Combined real-time PCR and rpoB gene pyrosequencing for rapid identification of Mycobacterium tuberculosis and determination of rifampin resistance directly in clinical specimens. J Clin Microbiol. 2010;48:1182–8. - PMC - PubMed
-
- Updated guidelines for the use of nucleic acid amplification tests in the diagnosis of tuberculosis. MMWR Morbid Mortal Wkly Rep. 2009;58(01):7–10. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

