A new V-ATPase regulatory mechanism mediated by the Rab interacting lysosomal protein (RILP)
- PMID: 26843904
- PMCID: PMC4594554
- DOI: 10.4161/cib.29616
A new V-ATPase regulatory mechanism mediated by the Rab interacting lysosomal protein (RILP)
Abstract
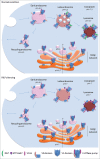
Progressive luminal acidification of intracellular compartments is important for their functions. Proton transport into the organelle's lumen is mediated by vacuolar ATPases (V-ATPases) large multi-subunit proton pumps organized into 2 domains, V0 and V1, working together as a rotary machine. The interaction of each subunit with specific partners plays a crucial role in controlling V-ATPase activity. Recently, we have shown that RILP, a Rab7 effector regulating late endocytic traffic and biogenesis of multivesicular bodies (MVBs), is a specific interactor of the V-ATPase subunit V1G1, a fundamental component of the peripheral stalk for correct V-ATPase assembly. RILP controls V1G1 stability and localization affecting V-ATPase assembly and function at the level of endosomes and lysosomes. The discovery of this new regulatory mechanism for V-ATPase opens new scenario to the comprehension of organelle's pH regulation and reveals a key role of RILP in controlling different aspects of endosome to lysosome transport.
Keywords: Endocytosis; Membrane traffic; RILP; Rab proteins; Rab7; Ubiquitin; V-ATPase; V1G1; atp6v1g1.
Figures
Similar articles
-
Role of the V1G1 subunit of V-ATPase in breast cancer cell migration.Sci Rep. 2021 Feb 25;11(1):4615. doi: 10.1038/s41598-021-84222-9. Sci Rep. 2021. PMID: 33633298 Free PMC article.
-
RILP regulates vacuolar ATPase through interaction with the V1G1 subunit.J Cell Sci. 2014 Jun 15;127(Pt 12):2697-708. doi: 10.1242/jcs.142604. Epub 2014 Apr 24. J Cell Sci. 2014. PMID: 24762812
-
Collapse of late endosomal pH elicits a rapid Rab7 response via the V-ATPase and RILP.J Cell Sci. 2024 May 1;137(9):jcs261765. doi: 10.1242/jcs.261765. Epub 2024 May 13. J Cell Sci. 2024. PMID: 38578235 Free PMC article.
-
The emerging roles of vacuolar-type ATPase-dependent Lysosomal acidification in neurodegenerative diseases.Transl Neurodegener. 2020 May 11;9(1):17. doi: 10.1186/s40035-020-00196-0. Transl Neurodegener. 2020. PMID: 32393395 Free PMC article. Review.
-
Structure and properties of the clathrin-coated vesicle and yeast vacuolar V-ATPases.J Bioenerg Biomembr. 1999 Feb;31(1):57-65. doi: 10.1023/a:1005496530380. J Bioenerg Biomembr. 1999. PMID: 10340849 Review.
Cited by
-
Role of the V1G1 subunit of V-ATPase in breast cancer cell migration.Sci Rep. 2021 Feb 25;11(1):4615. doi: 10.1038/s41598-021-84222-9. Sci Rep. 2021. PMID: 33633298 Free PMC article.
-
USP4 depletion-driven RAB7A ubiquitylation impairs autophagosome-lysosome fusion and aggravates periodontitis.Autophagy. 2025 Apr;21(4):771-788. doi: 10.1080/15548627.2024.2429371. Epub 2024 Dec 11. Autophagy. 2025. PMID: 39663592 Free PMC article.
-
Role of the RAB7 Protein in Tumor Progression and Cisplatin Chemoresistance.Cancers (Basel). 2019 Aug 1;11(8):1096. doi: 10.3390/cancers11081096. Cancers (Basel). 2019. PMID: 31374919 Free PMC article. Review.
-
Cln1-mutations suppress Rab7-RILP interaction and impair autophagy contributing to neuropathology in a mouse model of infantile neuronal ceroid lipofuscinosis.J Inherit Metab Dis. 2020 Sep;43(5):1082-1101. doi: 10.1002/jimd.12242. Epub 2020 Apr 27. J Inherit Metab Dis. 2020. PMID: 32279353 Free PMC article.
-
The Rab7 subfamily across Paramecium aurelia species; evidence of high conservation in sequence and function.Small GTPases. 2020 Nov;11(6):421-429. doi: 10.1080/21541248.2018.1502056. Epub 2018 Aug 29. Small GTPases. 2020. PMID: 30156960 Free PMC article.
References
-
- Casey JR, Grinstein S, Orlowski J. Sensors and regulators of intracellular pH. Nat Rev Mol Cell Biol 2010; 11:50-61; PMID:19997129; http://dx.doi.org/10.1038/nrm2820 - DOI - PubMed
-
- Mindell JA. Lysosomal acidification mechanisms. Annu Rev Physiol 2012; 74:69-86; PMID:22335796; http://dx.doi.org/10.1146/annurev-physiol-012110-142317 - DOI - PubMed
-
- Forgac M. Vacuolar ATPases: rotary proton pumps in physiology and pathophysiology. Nat Rev Mol Cell Biol 2007; 8:917-29; PMID:17912264; http://dx.doi.org/10.1038/nrm2272 - DOI - PubMed
-
- Marshansky V, Futai M. The V-type H+-ATPase in vesicular trafficking: targeting, regulation and function. Curr Opin Cell Biol 2008; 20:415-26; PMID:18511251; http://dx.doi.org/10.1016/j.ceb.2008.03.015 - DOI - PMC - PubMed
-
- Aniento F, Gu F, Parton RG, Gruenberg J. An endosomal β COP is involved in the pH-dependent formation of transport vesicles destined for late endosomes. J Cell Biol 1996; 133:29-41; PMID:8601610; http://dx.doi.org/10.1083/jcb.133.1.29 - DOI - PMC - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources
