Emergence of two prion subtypes in ovine PrP transgenic mice infected with human MM2-cortical Creutzfeldt-Jakob disease prions
- PMID: 26847207
- PMCID: PMC4743415
- DOI: 10.1186/s40478-016-0284-9
Emergence of two prion subtypes in ovine PrP transgenic mice infected with human MM2-cortical Creutzfeldt-Jakob disease prions
Abstract
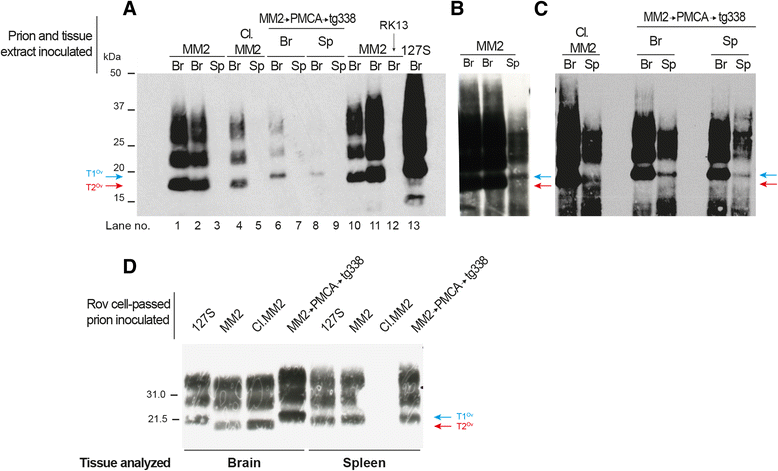
Introduction: Mammalian prions are proteinaceous pathogens responsible for a broad range of fatal neurodegenerative diseases in humans and animals. These diseases can occur spontaneously, such as Creutzfeldt-Jakob disease (CJD) in humans, or be acquired or inherited. Prions are primarily formed of macromolecular assemblies of the disease-associated prion protein PrP(Sc), a misfolded isoform of the host-encoded prion protein PrP(C). Within defined host-species, prions can exist as conformational variants or strains. Based on both the M/V polymorphism at codon 129 of PrP and the electrophoretic signature of PrP(Sc) in the brain, sporadic CJD is classified in different subtypes, which may encode different strains. A transmission barrier, the mechanism of which remains unknown, limits prion cross-species propagation. To adapt to the new host, prions have the capacity to 'mutate' conformationally, leading to the emergence of a variant with new biological properties. Here, we transmitted experimentally one rare subtype of human CJD, designated cortical MM2 (129 MM with type 2 PrP(Sc)), to transgenic mice overexpressing either human or the VRQ allele of ovine PrP(C).
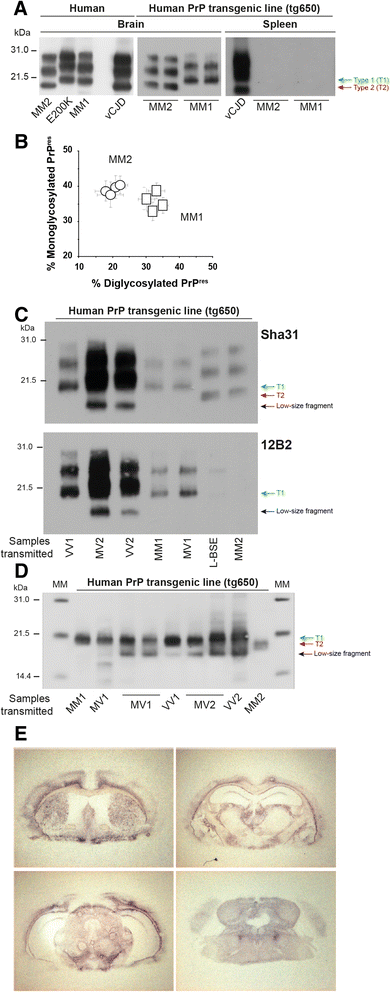
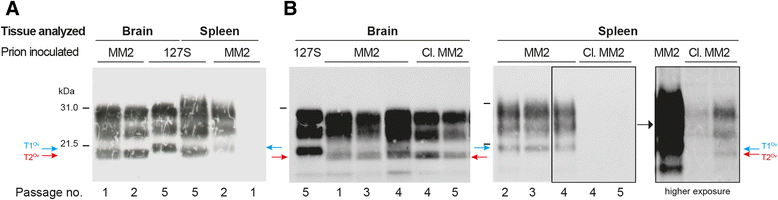
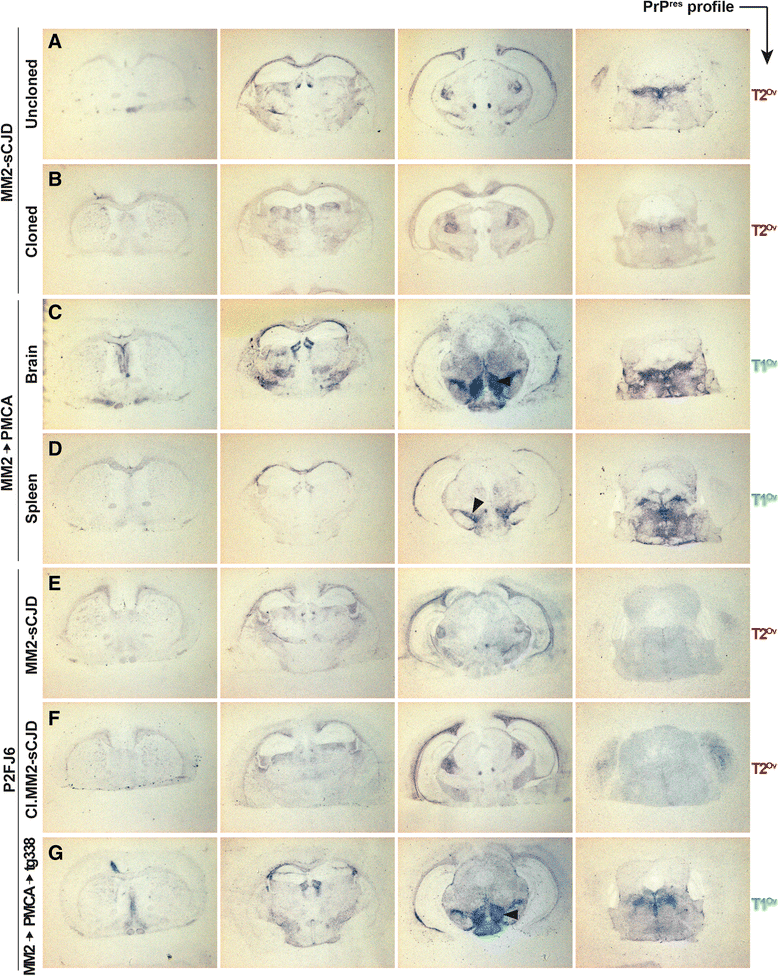
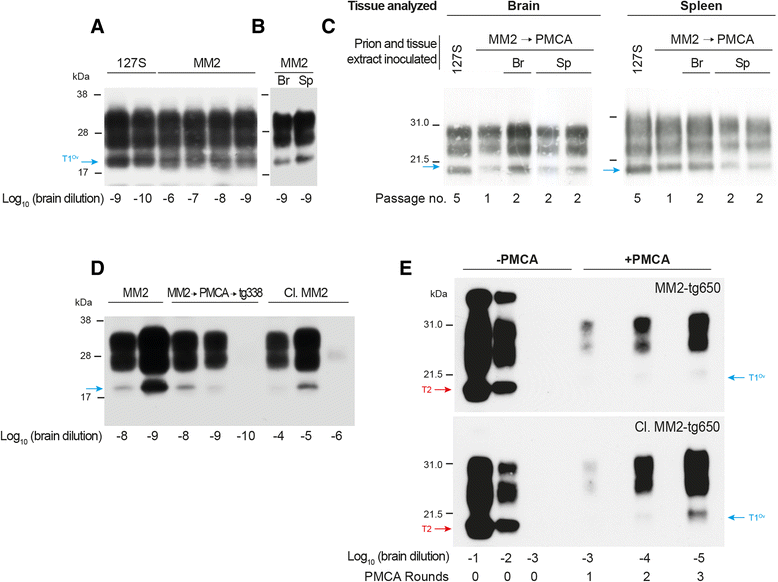
Results: In marked contrast with the reported absence of transmission to knock-in mice expressing physiological levels of human PrP, this subtype transmitted faithfully to mice overexpressing human PrP, and exhibited unique strain features. Onto the ovine PrP sequence, the cortical MM2 subtype abruptly evolved on second passage, thereby allowing emergence of a pair of strain variants with distinct PrP(Sc) biochemical characteristics and differing tropism for the central and lymphoid tissues. These two strain components exhibited remarkably distinct replicative properties in cell-free amplification assay, allowing the 'physical' cloning of the minor, lymphotropic component, and subsequent isolation in ovine PrP mice and RK13 cells.
Conclusions: Here, we provide in-depth assessment of the transmissibility and evolution of one rare subtype of sporadic CJD upon homologous and heterologous transmission. The notion that the environment or matrix where replication is occurring is key to the selection and preferential amplification of prion substrain components raises new questions on the determinants of prion replication within and between species. These data also further interrogate on the interplay between animal and human prions.
Figures
Similar articles
-
Prions from Sporadic Creutzfeldt-Jakob Disease Patients Propagate as Strain Mixtures.mBio. 2020 Jun 16;11(3):e00393-20. doi: 10.1128/mBio.00393-20. mBio. 2020. PMID: 32546613 Free PMC article.
-
Absence of Evidence for a Causal Link between Bovine Spongiform Encephalopathy Strain Variant L-BSE and Known Forms of Sporadic Creutzfeldt-Jakob Disease in Human PrP Transgenic Mice.J Virol. 2016 Nov 14;90(23):10867-10874. doi: 10.1128/JVI.01383-16. Print 2016 Dec 1. J Virol. 2016. PMID: 27681129 Free PMC article.
-
A traceback phenomenon can reveal the origin of prion infection.Neuropathology. 2009 Oct;29(5):619-24. doi: 10.1111/j.1440-1789.2008.00973.x. Epub 2009 Jul 29. Neuropathology. 2009. PMID: 19659941
-
Neuropathological and biochemical criteria to identify acquired Creutzfeldt-Jakob disease among presumed sporadic cases.Neuropathology. 2016 Jun;36(3):305-10. doi: 10.1111/neup.12270. Epub 2015 Dec 15. Neuropathology. 2016. PMID: 26669818 Review.
-
Prion encephalopathies of animals and humans.Dev Biol Stand. 1993;80:31-44. Dev Biol Stand. 1993. PMID: 8270114 Review.
Cited by
-
Two distinct conformers of PrPD type 1 of sporadic Creutzfeldt-Jakob disease with codon 129VV genotype faithfully propagate in vivo.Acta Neuropathol Commun. 2021 Mar 25;9(1):55. doi: 10.1186/s40478-021-01132-7. Acta Neuropathol Commun. 2021. PMID: 33766126 Free PMC article.
-
Prion strains associated with iatrogenic CJD in French and UK human growth hormone recipients.Acta Neuropathol Commun. 2021 Aug 28;9(1):145. doi: 10.1186/s40478-021-01247-x. Acta Neuropathol Commun. 2021. PMID: 34454616 Free PMC article.
-
A stretch of residues within the protease-resistant core is not necessary for prion structure and infectivity.Prion. 2017 Jan 2;11(1):25-30. doi: 10.1080/19336896.2016.1274851. Epub 2017 Feb 8. Prion. 2017. PMID: 28281924 Free PMC article.
-
Divergent prion strain evolution driven by PrPC expression level in transgenic mice.Nat Commun. 2017 Jan 23;8:14170. doi: 10.1038/ncomms14170. Nat Commun. 2017. PMID: 28112164 Free PMC article.
-
PrPC Governs Susceptibility to Prion Strains in Bank Vole, While Other Host Factors Modulate Strain Features.J Virol. 2016 Nov 14;90(23):10660-10669. doi: 10.1128/JVI.01592-16. Print 2016 Dec 1. J Virol. 2016. PMID: 27654300 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

