Antioxidant treatments for schizophrenia
- PMID: 26848926
- PMCID: PMC10337514
- DOI: 10.1002/14651858.CD008919.pub2
Antioxidant treatments for schizophrenia
Abstract
Background: There is accumulating evidence that progressive changes in brain structure and function take place as schizophrenia unfolds. Among many possible candidates, oxidative stress may be one of the mediators of neuroprogression, grey matter loss and subsequent cognitive and functional impairment. Antioxidants are exogenous or endogenous molecules that mitigate any form of oxidative stress or its consequences. They may act from directly scavenging free radicals to increasing anti-oxidative defences. There is evidence that current treatments impact oxidative pathways and may to some extent reverse pro-oxidative states in schizophrenia. The existing literature, however, indicates that these treatments do not fully restore the deficits in antioxidant levels or restore levels of oxidants in schizophrenia. As such, there has been interest in developing interventions aimed at restoring this oxidative balance beyond the benefits of antipsychotics in this direction. If antioxidants are to have a place in the treatment of this serious condition, the relevant and up-to-date information should be available to clinicians and investigators.
Objectives: To evaluate the effect of antioxidants as add-on treatments to standard antipsychotic medication for improving acute psychotic episodes and core symptoms, and preventing relapse in people with schizophrenia.
Search methods: We searched the Cochrane Schizophrenia Group's Study-Based Register of Trials which is based on regular searches of CINAHL, BIOSIS, AMED, Embase, PubMed, MEDLINE, PsycINFO, and registries of clinical trials. There are no language, time, document type, or publication status limitations for inclusion of records in the register. We ran this search in November 2010, and again on 8 January 2015. We also inspected references of all identified studies for further trials and contacted authors of trials for additional information.
Selection criteria: We included reports if they were randomised controlled trials (RCTs) involving people with schizophrenia who had been allocated to either a substance with antioxidant potential or to a placebo as an adjunct to standard antipsychotic treatment.
Data collection and analysis: We independently extracted data from these trials and we estimated risk ratios (RR) or mean differences (MD), with 95% confidence intervals (CI). We assessed risk of bias for included studies and created a 'Summary of findings' table using GRADE.
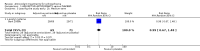
Main results: The review includes 22 RCTs of varying quality and sample size studying Ginkgo biloba, N-acetyl cysteine (NAC), allopurinol, dehydroepiandrosterone (DHEA), vitamin C, vitamin E or selegiline. Median follow-up was eight weeks. Only three studies including a minority of the participants reported our a priori selected primary outcome of clinically important response. Short-term data for this outcome (measured as at least 20% improvement in scores on Positive and Negative Syndrome Scale (PANSS)) were similar (3 RCTs, n = 229, RR 0.77, 95% CI 0.53 to 1.12, low quality evidence). Studies usually reported only endpoint psychopathology rating scale scores. Psychotic symptoms were lower in those using an adjunctive antioxidant according to the PANSS ( 7 RCTS, n = 584, MD -6.00, 95% CI -10.35 to -1.65, very low quality evidence) and the Brief Psychiatric Rating Scale (BPRS) (8 RCTS, n = 843, MD -3.20, 95% CI -5.63 to -0.78, low quality evidence). There was no overall short-term difference in leaving the study early (16 RCTs, n = 1584, RR 0.73, 95% CI 0.48 to 1.11, moderate quality evidence), or in general functioning (2 RCTs, n = 52, MD -1.11, 95% CI -8.07 to 5.86, low quality evidence). Adverse events were generally poorly reported. Three studies reported useable data for 'any serious adverse effect', results were equivocal (3 RCTs, n = 234, RR 0.65, 95% CI 0.19 to 2.27, low quality evidence). No evidence was available for relapse, quality of life or service use.
Authors' conclusions: Although 22 trials could be included in this review, the evidence provided is limited and mostly not relevant to clinicians or consumers. Overall, although there was low risk of attrition and selective data reporting bias within the trials, the trials themselves were not adequately powered and need more substantial follow-up periods. There is a need for larger trials with longer periods of follow-up to be conducted. Outcomes should be meaningful for those with schizophrenia, and include measures of improvement and relapse (not just rating scale scores), functioning and quality of life and acceptability and, importantly, safety data.
Conflict of interest statement
Pedro Magalhães has no conflicts of interest. Olivia Dean has received grant support from the Brain and Behavior Foundation, Simons Autism Foundation, Stanley Medical Research Institute, Lilly, NHMRC and an ASBD/Servier grant. Anna Andreazza has no conflicts of interest. Michael Berk was the lead investigator for one potentially relevant study for the review (Berk 2008; Berk 2008a) , but did not extract data from these trials for this review. He has received grant/research support from the Stanley Medical Research Foundation, MBF, NHMRC, beyondblue, Geelong Medical Research Foundation, Bristol Myers Squibb, Eli Lilly, Glaxo SmithKline, Organon, Novartis, Mayne Pharma, Servier; has been a consultant/speaker for: Astra Zeneca, Bristol Myers Squibb, Eli Lilly, Glaxo SmithKline, Janssen Cilag, Lundbeck, Pfizer, Sanofi Synthelabo, Servier, Solvay, Wyeth. Professor Berk is co‐inventor on two provisional patents regarding the use of NAC and related compounds for psychiatric indications, which, whilst assigned to the Mental Health Research Institute, could lead to personal remuneration upon a commercialisation event. Flávio Kapczinski has received grant/research support from Astra‐Zeneca, Eli Lilly, the Janssen‐Cilag, Servier, CNPq, CAPES, NARSAD and the Stanley Medical Research Institute; has been a member of the speakers’ boards for Astra‐Zeneca, Eli Lilly, Janssen and Servier; and has served as a consultant for Servier.
Figures























Comment in
-
[Antioxidants as adjunctive therapy in schizophrenia].Ugeskr Laeger. 2016 May 16;178(20):V68238. Ugeskr Laeger. 2016. PMID: 27189103 Danish. No abstract available.
References
References to studies included in this review
Adler 1993a {published data only}
-
- Adler LA, Peselow E, Rotrosen J, Duncan E, Lee M, Rosenthal M, et al. Vitamin E treatment of tardive dyskinesia. American Journal of Psychiatry 1993;150(9):1405‐7. - PubMed
-
- Adler LA, Edson R, Lavori P, Peselow E, Duncan E, Rosenthal M, et al. Long‐term treatment effects of vitamin E for tardive dyskinesia. Biological Psychiatry 1998;15:868‐72. - PubMed
Adler 1999 {published data only}
-
- Adler LA, Rotrosen J, Edson R, Lavori P, Lohr J, Hitzemann R, et al. Vitamin E treatment for tardive dyskinesia. Archives of General Psychiatry 1999;56(9):836‐41. [MEDLINE: ] - PubMed
Akhondzadeh 2005 {published data only}
-
- Akhondzadeh S, Safarcherati A, Amini H. Beneficial antipsychotic effects of allopurinol as add‐on therapy for schizophrenia: a double blind, randomized and placebo controlled trial. Progress in Neuro‐Psychopharmacology & Biological Psychiatry 2005;29(2):253‐9. [PUBMED: 15694232] - PubMed
Amiri 2008 {published data only}
-
- Amiri A, Noorbala AA, Nejatisafa AA, Ghoreishi A, Derakhshan MK, Khodaie‐Ardakani MR, et al. Efficacy of selegiline add on therapy to risperidone in the treatment of the negative symptoms of schizophrenia: a double‐blind randomized placebo‐controlled study. Human Psychopharmacology 2008;23(2):79‐86. [MEDLINE: ] - PubMed
Berk 2008b {published data only}
-
- Berk M, Copolov D, Dean O, Lu K, Jeavons S, Schapkaitz I, et al. N‐acetyl cysteine as a glutathione precursor for schizophrenia‐a double‐blind, randomized, placebo‐controlled trial. Biological Psychiatry 2008;64(5):361‐8. - PubMed
Bodkin 2005 {published data only}
-
- Bodkin JA, Siris SG, Bermanzohn PC, Hennen J, Cole JO. Double‐blind, placebo‐controlled, multicenter trial of selegiline augmentation of antipsychotic medication to treat negative symptoms in outpatients with schizophrenia. American Journal of Psychiatry 2005;162(2):388‐90. [MEDLINE: ] - PubMed
Bordbar 2008 {published data only}
-
- Bordbar MR, Abdollahian E, Hojjat K, Samaric A. Effects of selegiline on negative symptoms in schizophrenia: A double‐blind clinical trial. Iranian Journal of Psychiatry and Clinical Psychology 2008;14(2):131‐9.
Brunstein 2005 {published data only}
-
- Brunstein MG, Ghisolfi ES, Ramos FL, Lara DR. A clinical trial of adjuvant allopurinol therapy for moderately refractory schizophrenia. Journal of Clinical Psychiatry 2005;66(2):213‐9. [PUBMED: 15705007] - PubMed
Dakhale 2005 {published data only}
-
- Dakhale GN, Khanzode SD, Khanzode SS, Saoji A. Supplementation of vitamin C with atypical antipsychotics reduces oxidative stress and improves the outcome of schizophrenia. Psychopharmacology 2005;182(4):494‐8. [MEDLINE: ] - PubMed
Dickerson 2009 {published data only}
-
- Dickerson FB, Stallings CR, Origoni AE, Sullens A, Khushalani S, Sandson N, et al. A double‐blind trial of adjunctive allopurinol for schizophrenia. Schizophrenia Research 2009;109(1‐3):66‐9. [PUBMED: 19195842] - PubMed
Dorevitch 1997a {published data only}
-
- Dorevitch A, Kalian M, Shlafman M, Lerner V. Treatment of long‐term tardive dyskinesia with vitamin E. Biological Psychiatry 1997;41(1):114‐6. [MEDLINE: ] - PubMed
Doruk 2008 {published data only}
-
- Doruk A, Uzun O, Ozsahin A. A placebo‐controlled study of extract of ginkgo biloba added to clozapine in patients with treatment‐resistant schizophrenia. International Clinical Psychopharmacology 2008;23(4):223‐7. [PUBMED: 18545061] - PubMed
Farokhnia 2013 {published data only}
-
- Farokhnia M, Azarkolah A, Adinehfar F, Khodaie‐Ardakani MR, Hosseini SM, Yekehtaz H, et al. N‐acetylcysteine as an adjunct to risperidone for treatment of negative symptoms in patients with chronic schizophrenia: a randomized, double‐blind, placebo‐controlled study. Clinical Neuropharmacology 2013;36(6):185‐92. [PUBMED: 24201233] - PubMed
Goff 1993 {published data only}
-
- Goff DC, Renshaw PF, SaridSegal O, Dreyfuss DA, Amico ET, Ciraulo DA. A placebo‐controlled trial of selegiline (L‐deprenyl) in the treatment of tardive dyskinesia. Biological Psychiatry 1993;33(10):700‐6. [MEDLINE: ] - PubMed
Jungerman 1999 {published data only}
-
- Jungerman T, Rabinowitz D, Klein E. Deprenyl augmentation for treating negative symptoms of schizophrenia: a double blind, controlled study. Journal of Clinical Psychopharmacology 1999;19(6):522‐5. [MEDLINE: ] - PubMed
Lohr 1996 {published data only}
-
- Lohr JB, Caligiuri MP. A double‐blind placebo‐controlled study of vitamin E treatment of tardive dyskinesia. Journal of Clinical Psychiatry 1996;57(4):167‐73. - PubMed
Luo 1997 {published data only}
-
- Luo HC, Shen YC, Meng FQ. Therapeutic effect of shuxuening combining neuroleptics for the treatment of chronic schizophrenia‐‐a double blind study. Zhongguo Zhong xi yi jie he za zhi Zhongguo Zhongxiyi jiehe zazhi = Chinese Journal of Integrated Traditional and Western Medicine / Zhongguo Zhong xi yi jie he xue hui, Zhongguo Zhong yi yan jiu yuan zhu ban 1997;17(3):139‐42. - PubMed
Ritsner 2010 {published data only}
-
- Ritsner MS, Gibel A, Shleifer T, Boguslavsky I, Zayed A, Maayan R, et al. Pregnenolone and dehydroepiandrosterone as an adjunctive treatment in schizophrenia and schizoaffective disorder: an 8‐week, double‐blind, randomized, controlled, 2‐center, parallel‐group trial. The Journal of Clinical Psychiatry 2010;71(10):1351‐62. [PUBMED: 20584515] - PubMed
Weiser 2012 {published data only}
-
- Weiser M, Gershon AA, Rubinstein K, Petcu C, Ladea M, Sima D, et al. A randomized controlled trial of allopurinol vs. placebo added on to antipsychotics in patients with schizophrenia or schizoaffective disorder. Schizophrenia Research 2012;138(1):35‐8. [PUBMED: 22483162] - PubMed
Zhang 2001a {published data only}
-
- Zhang XY, Zhou DF, Zhang PY, Wu GY, Su JM, Cao LY. A double‐blind, placebo‐controlled trial of extract of Ginkgo biloba added to haloperidol in treatment‐resistant patients with schizophrenia. Journal of Clinical Psychiatry 2001;62(11):878‐83. [PUBMED: 11775047] - PubMed
Zhang 2004 {published data only}
-
- Zhang XY, Zhou DF, Cao LY, Xu CQ, Chen da C, Wu GY. The effect of vitamin e treatment on tardive dyskinesia and blood superoxide dismutase: a double‐blind placebo‐controlled trial. Journal of Clinical Psychopharmacology 2004;24(1):83‐6. [MEDLINE: ] - PubMed
Zhang 2011 {published data only}
-
- Zhang WF, Tan YL, Zhang XY, Chan RC, Wu HR, Zhou DF. Extract of Ginkgo biloba treatment for tardive dyskinesia in schizophrenia: a randomized, double‐blind, placebo‐controlled trial. Journal of Clinical Psychiatry 2011;72(5):615‐21. [PUBMED: 20868638] - PubMed
-
- Zhang XY, Zhou DF, Su JM, Zhang PY. The effects of extract of ginkgo biloba added to haloperidol on superoxide dismutase in inpatients with chronic schizophrenia. Journal of Clinical Psychopharmacology 2001;21(1):85‐8. - PubMed
References to studies excluded from this review
Bhavani 1962 {published data only}
-
- Bhavani AD, Sen NN, Punekar BD. Impact of ascorbic acid administration on memory, attention, intelligence and motor functions in schizophrenics. Transactions of all India Institute of Mental Health 1962;3:80‐8.
Dorevitch 1997 {published data only}
-
- Dorevitch A, Lerner V, Shalfman M, Kalian M. Lack of effect of vitamin E on serum creatine phosphokinase in patients with long‐term tardive dyskinesia. International Clinical Psychopharmacology 1997;12(3):171‐3. [PUBMED: 9248874] - PubMed
Elkashef 1990 {published data only}
-
- Elkashef AM, Ruskin PE, Bacher N, Barrett D. Vitamin E in the treatment of tardive dyskinesia. American Journal of Psychiatry 1990;147(4):505‐6. - PubMed
Eranti 1998 {published data only}
-
- Eranti VS, Gangadhar BN, Janakiramaiah N. Haloperidol induced extrapyramidal reaction: lack of protective effect by vitamin E. Psychopharmacology 1998;140(4):418‐20. [MEDLINE: ] - PubMed
Kapur 1991 {published data only}
-
- Kapur S, Ganguli R, Ulrich R, Raghu U. Use of random‐sequence riboflavin as a marker of medication compliance in chronic schizophrenics. Schizophrenia Research 1991;6(1):49‐53. [MEDLINE: ] - PubMed
Rees 1951 {published data only}
-
- Rees L, King GM. Desoxycortone acetate and ascorbic acid in the treatment of schizophrenia. Journal of Mental Science 1951;97:376‐80. - PubMed
Suresh 2007 {published data only}
-
- Suresh Kumar PN, Andrade C, Bhakta SG, Singh NM. Melatonin in schizophrenic outpatients with insomnia: a double‐blind, placebo‐controlled study. Journal of Clinical Psychiatry 2007;68(2):237‐41. [PUBMED: 17335321] - PubMed
References to studies awaiting assessment
Akhtar 1993 {published data only}
-
- Akhtar S, Jajor TR, Kumar S. Vitamin E in the treatment of tardive dyskinesia. Journal of Postgraduate Medicine 1993;39(3):124‐6. [MEDLINE: ] - PubMed
Altman 1973 {published data only}
-
- Altman H, Mehta D, Evenson RC, Sletten IW. Behavioral effects of drug therapy on psychogeriatric inpatients: II. multivitamin supplement. Journal of the American Geriatrics Society 1973;21(6):249‐52. [MEDLINE: ] - PubMed
Atmaca 2005 {published data only}
-
- Atmaca M, Tezcan E, Kuloglu M, Ustundag B, Kirtas O. The effect of extract of ginkgo biloba addition to olanzapine on therapeutic effect and antioxidant enzyme levels in patients with schizophrenia. Psychiatry and Clinical Neurosciences 2005;59(6):652‐6. [MEDLINE: ] - PubMed
Ben‐Dor 1998 {published data only}
-
- Ben‐Dor A, Gelkoph M. Vitamin E: an alternative to anticholinergic drugs?. Proceedings of the 9th Congress of the Association of European Psychiatrists; 1998 Sep 20‐24; Copenhagen, Denmark. 1998.
Egan 1992 {published data only}
-
- Egan MF, Hyde TM, Albers GW, Elkashef A, Alexander RC, Reeve A, et al. Treatment of tardive dyskinesia with vitamin E. American Journal of Psychiatry 1992;149(6):773‐7. - PubMed
Junker 1992 {published data only}
-
- Junker D, Steigleider P, Gattaz WF. Alpha‐tocopherol in the treatment of tardive dyskinesia. Proceedings of the 7th Biennial Winter Workshop on Schizophrenia; 1994 Jan 23‐28; Les Diablerets, Switzerland. 1992; Vol. 6, issue 2:123.
Kodesh 2003 {published data only}
-
- Kodesh A, Weizman A, Aizenberg D, Hermesh H, Gelkopf M, Zemishlany Z. Selegiline in the treatment of sexual dysfunction in schizophrenic patients maintained on neuroleptics: a pilot study. Clinical Neuropharmacology 2003;26(4):193‐5. [MEDLINE: ] - PubMed
Lam 1994 {published data only}
-
- Lam LCW, Chiu HFK, Hung SF. Vitamin E in the treatment of tardive dyskinesia: A replication study. Journal of Nervous and Mental Disease 1994;182(2):113‐4. - PubMed
Meng 1996 {published data only}
-
- Meng Fan Qiang, Cui Yu Hua, Wang Shan Hui. A double‐blind placebo controlled study of EGb761 in the treatment of chronic schizophrenia. Jounal of Clinical Psychological Medicine 1996;6:339‐341.
Milner 1963 {published data only}
-
- Milner G. Ascorbic acid in chronic psychiatric patients ‐ a controlled trial. British Journal of Psychiatry 1963;109:294‐9.
Salmasi 2009 {published data only}
-
- Salmasi FB, Jazayeri M, Ghaeli P, Hashemian F, Akhondzadeh S, Raisi F, et al. Comparing the effects of high‐dose vitamin E with those of placebo on insulin resistance in patients with schizophrenia treated with olanzapine. Journal of Clinical Psychopharmacology 2009;29(2):182‐3. [MEDLINE: ] - PubMed
Schmidt 1991 {published data only}
-
- Schmidt M, Meister P, Baumann P. Treatment of tardive dyskinesias with vitamin E. European Psychiatry 1991;6(4):201‐7.
Shamir 2000 {published data only}
-
- Shamir E, Barak Y, Plopsky I, Zisapel N, Elizur A, Weizman A. Is melatonin treatment effective for tardive dyskinesia?. Journal of Clinical Psychiatry 2000;61(8):556‐8. [MEDLINE: ] - PubMed
Shamir 2001a {published data only}
-
- Shamir E, Barak Y, Shalman I, Laudon M, Zisapel N, Tarrasch R, et al. Melatonin treatment for tardive dyskinesia: a double‐blind, placebo‐controlled, crossover study. Archives of General Psychiatry 2001;58:1049‐52. [MEDLINE: ] - PubMed
Shriqui 1992 {published data only}
-
- Shriqui CL, Bradwejn J, Annable L, Jones BD. Vitamin E in the treatment of tardive dyskinesia: a double‐blind placebo‐ controlled study. American Journal of Psychiatry 1992;149(3):391‐3. - PubMed
Stearns 1996 {published data only}
-
- Stearns AI, Sambunaris A, Elkashef AM, Issa F, Egan MF, Wyatt RJ. Selegiline for negative symptoms and tardive dyskinesia. Proceedings of the 149th Annual Meeting of the American Psychiatric Association; 1996 May 4‐9; New York, New York, USA. 1996.
Xu 2002 {published data only}
-
- Xu X, Ji Y, He M. The efficacy of Ginkgo biloba tablet inadjuvant treatment of schizophrenia. Shanghai Medical andPharmaceutical Journal 2002;23:456‐7.
Additional references
Altman 1996
Andreasen 1990
-
- Andreasen NC. Methods for assessing positive and negative symptoms. Modern problems of pharmacopsychiatry 1990;24:73‐88. [PUBMED: 2336066] - PubMed
Berk 2008
-
- Berk M, Copolov DL, Dean O, Lu K, Jeavons S, Schapkaitz I, et al. N‐acetyl cysteine for depressive symptoms in bipolar disorder ‐ a double‐blind randomized placebo‐controlled trial. Biological Psychiatry 2008;64(6):468‐75. [PUBMED: 18534556] - PubMed
Berk 2008a
-
- Berk M, Copolov DL, Dean O, Lu K, Jeavons S, Schapkaitz I, et al. N‐acetyl cysteine as a glutathione precursor for schizophrenia ‐ a double‐blind, randomized, placebo‐controlled trial. Biological Psychiatry 2008;64(5):361‐8. [PUBMED: 18436195] - PubMed
Berk 2009
-
- Berk M. Neuroprogression: pathways to progressive brain changes in bipolar disorder. International Journal of Neuropsychopharmacology 2009;12(4):441‐5. [PUBMED: 18922203] - PubMed
Boissel 1999
-
- Boissel JP, Cucherat M, Li W, Chatellier G, Gueyffier F, Buyse M, et al. The problem of therapeutic efficacy indices. 3. Comparison of the indices and their use [Apercu sur la problematique des indices d'efficacite therapeutique, 3: comparaison des indices et utilisation. Groupe d'Etude des Indices D'efficacite]. Therapie 1999;54(4):405‐11. [PUBMED: 10667106] - PubMed
Carpenter 1994
-
- Carpenter WT Jr, Buchanan RW. Schizophrenia. New England Journal of Medicine 1994;33(10):681‐90. - PubMed
Dean 2009
-
- Dean OM, Buuse M, Bush AI, Copolov DL, Ng F, Dodd S, et al. A role for glutathione in the pathophysiology of bipolar disorder and schizophrenia? Animal models and relevance to clinical practice. Current Medicinal Chemistry 2009;16(23):2965‐76. [PUBMED: 19689277] - PubMed
Deeks 2000
-
- Deeks J. Issues in the selection for meta‐analyses of binary data. Proceedings of the 8th International Cochrane Colloquium; 2000 Oct 25‐28; Cape town. Cape Town: The Cochrane Collaboration, 2000.
DeLisi 2008
DerSimonian 1986
-
- DerSimonian R, Laird N. Meta‐analysis in clinical trials. Controlled Clinical Trials 1986;7(3):177‐88. [PUBMED: 3802833] - PubMed
Egger 1997
Elbourne 2002
-
- Elbourne D, Altman DG, Higgins JPT, Curtina F, Worthingtond HV, Vaile A. Meta‐analyses involving cross‐over trials: methodological issues. International Journal of Epidemiology 2002;31(1):140‐9. - PubMed
Endicott 1976
-
- Endicott J, Spitzer RL, Fleiss JL, Cohen J. The global assessment scale. A procedure for measuring overall severity of psychiatric disturbance. Archives of General Psychiatry 1976;33(6):766‐71. [PUBMED: 938196] - PubMed
Freemantle 1999
-
- Freemantle N, Mason J, Eccles M. Deriving treatment recommendations from evidence within randomized trials. The role and limitation of meta‐analysis. International Journal of Technology Assessment in Health Care 1999;15(2):304‐15. [PUBMED: 10507190] - PubMed
Guy 1991
-
- Guy W. Treatment Emergent Symptom Scale. ECDEU Assessment Manual for Psychopharmacology. National Institutes for Mental Health, 1991.
Higgins 2003
Higgins 2011
-
- Higgins JPT, Green S (editors). Cochrane Handbook for Systematic Reviews of Interventions Version 5.0.2 [updated September 2011]. The Cochrane Collaboration, 2011. Available from www.cochrane‐handbook.org.
Hutton 2009
-
- Hutton JL. Number needed to treat and number needed to harm are not the best way to report and assess the results of randomised clinical trials. British Journal of Haematology 2009;146(1):27‐30. - PubMed
Jones 1995
-
- Jones SH, Thornicroft G, Coffey M, Dunn G. A brief mental health outcome scale‐reliability and validity of the Global Assessment of Functioning (GAF). British Journal of Psychiatry: the journal of mental science 1995;166(5):654‐9. [PUBMED: 7620753] - PubMed
Kay 1986
-
- Kay SR, Opler LA, Fiszbein A. Positive and negative syndrome scale (PANSS) manual. North Tonawanda, NY: Multi‐Health Systems, 1986.
Leucht 2005
-
- Leucht S, Kane JM, Kissling W, Hamann J, Etschel E, Engel R. Clinical implications of Brief Psychiatric Rating Scale scores. British Journal of Psychiatry 2005;187:366‐71. [PUBMED: 16199797] - PubMed
Leucht 2005a
-
- Leucht S, Kane JM, Kissling W, Hamann J, Etschel E, Engel RR. What does the PANSS mean?. Schizophrenia Research 2005;79(2‐3):231‐8. [PUBMED: 15982856] - PubMed
Leucht 2007
Lieberman 2005
-
- Lieberman JA, Tollefson GD, Charles C, Zipursky R, Sharma T, Kahn RS, Keefe RS, Green AI, Gur RE, McEvoy J, Perkins D, Hamer RM, Gu H, Tohen M, HGDH Study Group. Antipsychotic drug effects on brain morphology in first‐episode psychosis. Archives of General Psychiatry 2005;62(4):361‐70. [PUBMED: 15809403] - PubMed
Lieberman 2006
-
- Lieberman JA, Malaspina D, Jarskog LF. Preventing clinical deterioration in the course of schizophrenia: the potential for neuroprotection. CNS Spectrums 2006;11(4):Suppl 1‐13; Quiz Suppl 14‐5. [PUBMED: 16641837] - PubMed
Marshall 2000
-
- Marshall M, Lockwood A, Bradley C, Adams C, Joy C, Fenton M. Unpublished rating scales: a major source of bias in randomised controlled trials of treatments for schizophrenia. British Journal of Psychiatry 2000;176:249‐52. [PUBMED: 10755072] - PubMed
Ng 2008
-
- Ng F, Berk M, Dean O, Bush AI. Oxidative stress in psychiatric disorders: evidence base and therapeutic implications. International Journal of Neuropsychopharmacology 2008;11(6):851‐76. [PUBMED: 18205981] - PubMed
Overall 1962
-
- Overall JE, Gorham DR. The Brief Psychiatric Rating Scale. Psychological Reports 1962;10:799‐812.
Padurariu 2010
-
- Padurariu M, Ciobica A, Dobrin I, Stefanescu C. Evaluation of antioxidant enzymes activities and lipid peroxidation in schizophrenic patients treated with typical and atypical antipsychotics. Neuroscience Letters 2010;479(3):317‐20. [PUBMED: 20561936] - PubMed
Petiti 2000
-
- Petiti DB. Meta‐Analysis, Decision Analysis, and Cost‐Effectiveness Analysis: Methods for Quantitative Synthesis in Medicine. 2nd Edition. New York: Oxford University Press, 2000.
Rossler 2005
-
- Rossler W, Salize HJ, Os J, Riecher‐Rossler A. Size of burden of schizophrenia and psychotic disorders. European Neuropsychopharmacology 2005;15(4):399‐409. [PUBMED: 15925493] - PubMed
Satoh 1978
-
- Satoh K. Serum lipid peroxide in cerebrovascular disorders determined by a new colorimetric method. Clinica Chimica Acta; international journal of clinical chemistry 1978;90(1):37‐43. [PUBMED: 719890] - PubMed
Singh 2010
-
- Singh V, Singh SP, Chan K. Review and meta‐analysis of usage of ginkgo as an adjunct therapy in chronic schizophrenia. International Journal of Neuropsychopharmacology / official scientific journal of the Collegium Internationale Neuropsychopharmacologicum (CINP) 2010;13(2):257‐71. [PUBMED: 19775502] - PubMed
Soares‐Weiser 2011
Uttara 2009
Valko 2007
-
- Valko M, Leibfritz D, Moncol J, Cronin MT, Mazur M, Telser J. Free radicals and antioxidants in normal physiological functions and human disease. International journal of Biochemistry & Cell Biology 2007;39(1):44‐84. [PUBMED: 16978905] - PubMed
Wang 2008
Wood 2009
-
- Wood SJ, Yucel M, Pantelis C, Berk M. Neurobiology of schizophrenia spectrum disorders: the role of oxidative stress. Annals of the Academy of Medicine 2009;38(5):396‐6. [PUBMED: 19521638] - PubMed
Xia 2009
-
- Xia J, Adams CE, Bhagat N, Bhagat V, Bhoopathi P, El‐Sayeh H, et al. Loss to outcomes stakeholder survey: the LOSS study. Psychiatric Bulletin 2009;33(7):254‐7.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

