Human lamin B contains a farnesylated cysteine residue
- PMID: 2684976
- PMCID: PMC3443689
Human lamin B contains a farnesylated cysteine residue
Abstract
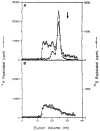
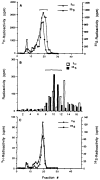
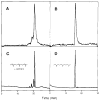
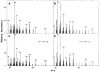
We recently showed that HeLa cell lamin B is modified by a mevalonic acid derivative. Here we identified the modified amino acid, determined its mode of linkage to the mevalonic acid derivative, and established the derivative's structure. A cysteine residue is modified because experiments with lamin B that had been biosynthetically labeled with [3H]mevalonic acid or [35S]cysteine and then extensively digested with proteases yielded 3H- or 35S-labeled products that co-chromatographed in five successive systems. A thioether linkage rather than a thioester linkage is involved because the mevalonic acid derivative could be released from the 3H-labeled products in a pentane-extractable form by treatment with Raney nickel but not with methanolic KOH. The derivative is a farnesyl moiety because the Raney nickel-released material was identified as 2,6,10-trimethyl-2,6,10-dodecatriene by a combination of gas chromatography and mass spectrometry. The thioether-modified cysteine residue appears to be located near the carboxyl end of lamin B because treatment of 3H-labeled lamin B with cyanogen bromide yielded a single labeled polypeptide that mapped toward this end of the cDNA-inferred sequence of human lamin B.
Figures

References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

