CO Metabolism in the Thermophilic Acetogen Thermoanaerobacter kivui
- PMID: 26850300
- PMCID: PMC4959504
- DOI: 10.1128/AEM.00122-16
CO Metabolism in the Thermophilic Acetogen Thermoanaerobacter kivui
Abstract
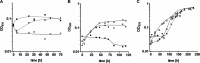
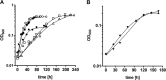
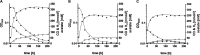
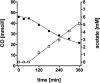
The thermophilic acetogenic bacterium Thermoanaerobacter kivui, previously described not to use carbon monoxide as a carbon and energy source, was adapted to grow on CO. This was achieved by using a preculture grown on H2 plus CO2 and by increasing the CO concentration in small, 10% increments.T. kivui was finally able to grow within a 100% CO atmosphere. Growth on CO was found in complex and mineral media, and vitamins were not required. Carbon monoxide consumption was accompanied by acetate and hydrogen production. Cells also grew on synthesis gas (syngas) with the simultaneous use of CO and H2 coupled to acetate production. CO oxidation in resting cells was coupled to hydrogen and acetate production and accompanied by the synthesis of ATP. A protonophore abolished ATP synthesis but stimulated H2 production, which is consistent with a chemiosmotic mechanism of ATP synthesis. Hydrogenase activity was highest in crude extracts of CO-grown cells, and carbon monoxide dehydrogenase (CODH) activity was highest in H2-plus-CO2- or CO-grown cells. The genome of T. kivui harbors two CODH gene clusters, and both CODH proteins were present in crude extracts, but one CODH was more prevalent in crude extracts from CO-grown cells.
Copyright © 2016, American Society for Microbiology. All Rights Reserved.
Figures


Similar articles
-
"Hot" acetogenesis.Extremophiles. 2017 Jan;21(1):15-26. doi: 10.1007/s00792-016-0873-3. Epub 2016 Sep 13. Extremophiles. 2017. PMID: 27623994 Review.
-
Energy conservation by a hydrogenase-dependent chemiosmotic mechanism in an ancient metabolic pathway.Proc Natl Acad Sci U S A. 2019 Mar 26;116(13):6329-6334. doi: 10.1073/pnas.1818580116. Epub 2019 Mar 8. Proc Natl Acad Sci U S A. 2019. PMID: 30850546 Free PMC article.
-
The energy-converting hydrogenase Ech2 is important for the growth of the thermophilic acetogen Thermoanaerobacter kivui on ferredoxin-dependent substrates.Microbiol Spectr. 2024 Apr 2;12(4):e0338023. doi: 10.1128/spectrum.03380-23. Epub 2024 Feb 22. Microbiol Spectr. 2024. PMID: 38385688 Free PMC article.
-
A genome-guided analysis of energy conservation in the thermophilic, cytochrome-free acetogenic bacterium Thermoanaerobacter kivui.BMC Genomics. 2014 Dec 18;15(1):1139. doi: 10.1186/1471-2164-15-1139. BMC Genomics. 2014. PMID: 25523312 Free PMC article.
-
Carbon monoxide. Toxic gas and fuel for anaerobes and aerobes: carbon monoxide dehydrogenases.Met Ions Life Sci. 2014;14:37-69. doi: 10.1007/978-94-017-9269-1_3. Met Ions Life Sci. 2014. PMID: 25416390 Review.
Cited by
-
"Hot" acetogenesis.Extremophiles. 2017 Jan;21(1):15-26. doi: 10.1007/s00792-016-0873-3. Epub 2016 Sep 13. Extremophiles. 2017. PMID: 27623994 Review.
-
DNA uptake from a laboratory environment drives unexpected adaptation of a thermophile to a minor medium component.ISME Commun. 2023 Jan 11;3(1):2. doi: 10.1038/s43705-022-00211-7. ISME Commun. 2023. PMID: 37938748 Free PMC article.
-
Lactate formation from fructose or C1 compounds in the acetogen Acetobacterium woodii by metabolic engineering.Appl Microbiol Biotechnol. 2023 Sep;107(17):5491-5502. doi: 10.1007/s00253-023-12637-7. Epub 2023 Jul 7. Appl Microbiol Biotechnol. 2023. PMID: 37417977 Free PMC article.
-
A Genetic System for the Thermophilic Acetogenic Bacterium Thermoanaerobacter kivui.Appl Environ Microbiol. 2018 Jan 17;84(3):e02210-17. doi: 10.1128/AEM.02210-17. Print 2018 Feb 1. Appl Environ Microbiol. 2018. PMID: 29150512 Free PMC article.
-
Genomic and in-situ Transcriptomic Characterization of the Candidate Phylum NPL-UPL2 From Highly Alkaline Highly Reducing Serpentinized Groundwater.Front Microbiol. 2018 Dec 18;9:3141. doi: 10.3389/fmicb.2018.03141. eCollection 2018. Front Microbiol. 2018. PMID: 30619209 Free PMC article.
References
-
- Sokolova TG, Gonzalez JM, Kostrikina NA, Chernyh NA, Slepova TV, Bonch-Osmolovskaya EA, Robb FT. 2004. Thermosinus carboxydivorans gen. nov., sp. nov., a new anaerobic, thermophilic, carbon-monoxide-oxidizing, hydrogenogenic bacterium from a hot pool of Yellowstone National Park. Int J Syst Evol Microbiol 54:2353–2359. doi:10.1099/ijs.0.63186-0. - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

