Integrated decision strategies for skin sensitization hazard
- PMID: 26851134
- PMCID: PMC4945438
- DOI: 10.1002/jat.3281
Integrated decision strategies for skin sensitization hazard
Erratum in
-
Integrated decision strategies for skin sensitization hazard.J Appl Toxicol. 2018 Mar;38(3):432. doi: 10.1002/jat.3572. Epub 2017 Nov 29. J Appl Toxicol. 2018. PMID: 29363186 No abstract available.
Abstract
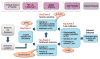
One of the top priorities of the Interagency Coordinating Committee for the Validation of Alternative Methods (ICCVAM) is the identification and evaluation of non-animal alternatives for skin sensitization testing. Although skin sensitization is a complex process, the key biological events of the process have been well characterized in an adverse outcome pathway (AOP) proposed by the Organisation for Economic Co-operation and Development (OECD). Accordingly, ICCVAM is working to develop integrated decision strategies based on the AOP using in vitro, in chemico and in silico information. Data were compiled for 120 substances tested in the murine local lymph node assay (LLNA), direct peptide reactivity assay (DPRA), human cell line activation test (h-CLAT) and KeratinoSens assay. Data for six physicochemical properties, which may affect skin penetration, were also collected, and skin sensitization read-across predictions were performed using OECD QSAR Toolbox. All data were combined into a variety of potential integrated decision strategies to predict LLNA outcomes using a training set of 94 substances and an external test set of 26 substances. Fifty-four models were built using multiple combinations of machine learning approaches and predictor variables. The seven models with the highest accuracy (89-96% for the test set and 96-99% for the training set) for predicting LLNA outcomes used a support vector machine (SVM) approach with different combinations of predictor variables. The performance statistics of the SVM models were higher than any of the non-animal tests alone and higher than simple test battery approaches using these methods. These data suggest that computational approaches are promising tools to effectively integrate data sources to identify potential skin sensitizers without animal testing. Published 2016. This article has been contributed to by US Government employees and their work is in the public domain in the USA.
Keywords: DPRA; KeratinoSens; LLNA; allergic contact dermatitis; h-CLAT; integrated decision strategy; machine learning; skin sensitization; support vector machine.
Published 2016. This article has been contributed to by US Government employees and their work is in the public domain in the USA.
Conflict of interest statement
The authors declare that there are no conflicts of interest.
Figures

Similar articles
-
Multivariate models for prediction of human skin sensitization hazard.J Appl Toxicol. 2017 Mar;37(3):347-360. doi: 10.1002/jat.3366. Epub 2016 Aug 2. J Appl Toxicol. 2017. PMID: 27480324 Free PMC article.
-
Prediction of skin sensitization potency using machine learning approaches.J Appl Toxicol. 2017 Jul;37(7):792-805. doi: 10.1002/jat.3424. Epub 2017 Jan 10. J Appl Toxicol. 2017. PMID: 28074598 Free PMC article.
-
Non-animal assessment of skin sensitization hazard: Is an integrated testing strategy needed, and if so what should be integrated?J Appl Toxicol. 2018 Jan;38(1):41-50. doi: 10.1002/jat.3479. Epub 2017 May 24. J Appl Toxicol. 2018. PMID: 28543848 Review.
-
Evaluation of combinations of in vitro sensitization test descriptors for the artificial neural network-based risk assessment model of skin sensitization.J Appl Toxicol. 2015 Nov;35(11):1333-47. doi: 10.1002/jat.3105. Epub 2015 Mar 30. J Appl Toxicol. 2015. PMID: 25824844
-
Non-animal methods to predict skin sensitization (II): an assessment of defined approaches *.Crit Rev Toxicol. 2018 May;48(5):359-374. doi: 10.1080/10408444.2018.1429386. Epub 2018 Feb 23. Crit Rev Toxicol. 2018. PMID: 29474122 Free PMC article. Review.
Cited by
-
Deriving a Continuous Point of Departure for Skin Sensitization Risk Assessment Using a Bayesian Network Model.Toxics. 2024 Jul 24;12(8):536. doi: 10.3390/toxics12080536. Toxics. 2024. PMID: 39195638 Free PMC article.
-
Multivariate models for prediction of human skin sensitization hazard.J Appl Toxicol. 2017 Mar;37(3):347-360. doi: 10.1002/jat.3366. Epub 2016 Aug 2. J Appl Toxicol. 2017. PMID: 27480324 Free PMC article.
-
Evaluation of Delayed-Type Hypersensitivity to Antineoplastic Drugs-An Overview.Cancers (Basel). 2023 Feb 14;15(4):1208. doi: 10.3390/cancers15041208. Cancers (Basel). 2023. PMID: 36831549 Free PMC article. Review.
-
Immunotoxicology: A brief history, current status and strategies for future immunotoxicity assessment.Curr Opin Toxicol. 2017 Aug;5:55-59. doi: 10.1016/j.cotox.2017.08.002. Curr Opin Toxicol. 2017. PMID: 28989989 Free PMC article. No abstract available.
-
Adverse effects in traditional and alternative toxicity tests.Regul Toxicol Pharmacol. 2024 Mar;148:105579. doi: 10.1016/j.yrtph.2024.105579. Epub 2024 Feb 1. Regul Toxicol Pharmacol. 2024. PMID: 38309424 Free PMC article.
References
-
- 16 CFR 1500.3. Title 16- Commercial Practices. Consumer Product Safety Commission; 2014.
-
- 29 CFR 1910.1200. Title 29- Labor. Occupational Safety and Health Administration; 2012. Occupational Safety and Health Standards.
-
- 40 CFR 158.500. Title 40- Protection of Environment. Environmental Protection Agency; 2012.
-
- 40 CFR 161.340. Title 40- Protection of Environment. Environmental Protection Agency; 2012.
-
- AltTox. [November 4, 2015];Toxicity Endpoints & Tests: Skin Sensitization. 2014 http://alttox.org/mapp/toxicity-endpoints-tests/skin-sensitization/
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

