Inflammation and its role in age-related macular degeneration
- PMID: 26852158
- PMCID: PMC4819943
- DOI: 10.1007/s00018-016-2147-8
Inflammation and its role in age-related macular degeneration
Abstract
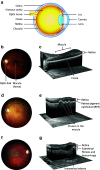
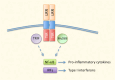
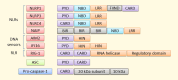
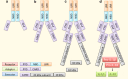
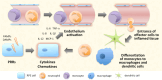
Inflammation is a cellular response to factors that challenge the homeostasis of cells and tissues. Cell-associated and soluble pattern-recognition receptors, e.g. Toll-like receptors, inflammasome receptors, and complement components initiate complex cellular cascades by recognizing or sensing different pathogen and damage-associated molecular patterns, respectively. Cytokines and chemokines represent alarm messages for leukocytes and once activated, these cells travel long distances to targeted inflamed tissues. Although it is a crucial survival mechanism, prolonged inflammation is detrimental and participates in numerous chronic age-related diseases. This article will review the onset of inflammation and link its functions to the pathogenesis of age-related macular degeneration (AMD), which is the leading cause of severe vision loss in aged individuals in the developed countries. In this progressive disease, degeneration of the retinal pigment epithelium (RPE) results in the death of photoreceptors, leading to a loss of central vision. The RPE is prone to oxidative stress, a factor that together with deteriorating functionality, e.g. decreased intracellular recycling and degradation due to attenuated heterophagy/autophagy, induces inflammation. In the early phases, accumulation of intracellular lipofuscin in the RPE and extracellular drusen between RPE cells and Bruch's membrane can be clinically detected. Subsequently, in dry (atrophic) AMD there is geographic atrophy with discrete areas of RPE loss whereas in the wet (exudative) form there is neovascularization penetrating from the choroid to retinal layers. Elevations in levels of local and systemic biomarkers indicate that chronic inflammation is involved in the pathogenesis of both disease forms.
Keywords: Aging; Eye; Immune system; NLRP3; Retina; Signaling.
Conflict of interest statement
The authors declare that they have no competing interests.
Figures
References
-
- Salvioli S, Capri M, Valensin S, Tieri P, Monti D, Ottaviani E, Franceschi C. Inflamm-aging, cytokines and aging: state of the art, new hypotheses on the role of mitochondria and new perspectives from systems biology. Curr Pharm Des. 2006;12:3161–3171. doi: 10.2174/138161206777947470. - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

