A proactive role of water molecules in acceptor recognition by protein O-fucosyltransferase 2
- PMID: 26854667
- PMCID: PMC4845761
- DOI: 10.1038/nchembio.2019
A proactive role of water molecules in acceptor recognition by protein O-fucosyltransferase 2
Abstract
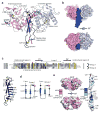
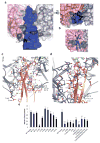
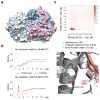
Protein O-fucosyltransferase 2 (POFUT2) is an essential enzyme that fucosylates serine and threonine residues of folded thrombospondin type 1 repeats (TSRs). To date, the mechanism by which this enzyme recognizes very dissimilar TSRs has been unclear. By engineering a fusion protein, we report the crystal structure of Caenorhabditis elegans POFUT2 (CePOFUT2) in complex with GDP and human TSR1 that suggests an inverting mechanism for fucose transfer assisted by a catalytic base and shows that nearly half of the TSR1 is embraced by CePOFUT2. A small number of direct interactions and a large network of water molecules maintain the complex. Site-directed mutagenesis demonstrates that POFUT2 fucosylates threonine preferentially over serine and relies on folded TSRs containing the minimal consensus sequence C-X-X-S/T-C. Crystallographic and mutagenesis data, together with atomic-level simulations, uncover a binding mechanism by which POFUT2 promiscuously recognizes the structural fingerprint of poorly homologous TSRs through a dynamic network of water-mediated interactions.
Conflict of interest statement
Figures

Similar articles
-
The Essential Role of Water Molecules in the Reaction Mechanism of Protein O-Fucosyltransferase 2.Angew Chem Int Ed Engl. 2022 Nov 25;61(48):e202213610. doi: 10.1002/anie.202213610. Epub 2022 Nov 10. Angew Chem Int Ed Engl. 2022. PMID: 36260536 Free PMC article.
-
O-fucosylation stabilizes the TSR3 motif in thrombospondin-1 by interacting with nearby amino acids and protecting a disulfide bond.J Biol Chem. 2022 Jun;298(6):102047. doi: 10.1016/j.jbc.2022.102047. Epub 2022 May 18. J Biol Chem. 2022. PMID: 35597280 Free PMC article.
-
Structure of human POFUT2: insights into thrombospondin type 1 repeat fold and O-fucosylation.EMBO J. 2012 Jul 18;31(14):3183-97. doi: 10.1038/emboj.2012.143. EMBO J. 2012. PMID: 22588082 Free PMC article.
-
Peters plus syndrome mutations disrupt a noncanonical ER quality-control mechanism.Curr Biol. 2015 Feb 2;25(3):286-295. doi: 10.1016/j.cub.2014.11.049. Epub 2014 Dec 24. Curr Biol. 2015. PMID: 25544610 Free PMC article.
-
A perspective on structural and mechanistic aspects of protein O-fucosylation.Acta Crystallogr F Struct Biol Commun. 2018 Aug 1;74(Pt 8):443-450. doi: 10.1107/S2053230X18004788. Epub 2018 Jul 26. Acta Crystallogr F Struct Biol Commun. 2018. PMID: 30084393 Free PMC article. Review.
Cited by
-
Protein O-fucosyltransferase 2-mediated O-glycosylation of the adhesin MIC2 is dispensable for Toxoplasma gondii tachyzoite infection.J Biol Chem. 2019 Feb 1;294(5):1541-1553. doi: 10.1074/jbc.RA118.005357. Epub 2018 Dec 4. J Biol Chem. 2019. PMID: 30514763 Free PMC article.
-
Protein O-Fucosyltransferase 2 Is Not Essential for Plasmodium berghei Development.Front Cell Infect Microbiol. 2019 Jul 3;9:238. doi: 10.3389/fcimb.2019.00238. eCollection 2019. Front Cell Infect Microbiol. 2019. PMID: 31334132 Free PMC article.
-
The Essential Role of Water Molecules in the Reaction Mechanism of Protein O-Fucosyltransferase 2.Angew Chem Int Ed Engl. 2022 Nov 25;61(48):e202213610. doi: 10.1002/anie.202213610. Epub 2022 Nov 10. Angew Chem Int Ed Engl. 2022. PMID: 36260536 Free PMC article.
-
Distinct C-mannosylation of netrin receptor thrombospondin type 1 repeats by mammalian DPY19L1 and DPY19L3.Proc Natl Acad Sci U S A. 2017 Mar 7;114(10):2574-2579. doi: 10.1073/pnas.1613165114. Epub 2017 Feb 15. Proc Natl Acad Sci U S A. 2017. PMID: 28202721 Free PMC article.
-
Structural insights into mechanism and specificity of the plant protein O-fucosyltransferase SPINDLY.Nat Commun. 2022 Dec 2;13(1):7424. doi: 10.1038/s41467-022-35234-0. Nat Commun. 2022. PMID: 36456586 Free PMC article.
References
-
- Hurtado-Guerrero R, Davies GJ. Recent structural and mechanistic insights into post-translational enzymatic glycosylation. Curr Opin Chem Biol. 2012;16:479–87. - PubMed
-
- Sakaidani Y, et al. O-linked-N-acetylglucosamine on extracellular protein domains mediates epithelial cell-matrix interactions. Nat Commun. 2011;2:583. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

