Immunohistochemistry for the detection of neural and inflammatory cells in equine brain tissue
- PMID: 26855862
- PMCID: PMC4741088
- DOI: 10.7717/peerj.1601
Immunohistochemistry for the detection of neural and inflammatory cells in equine brain tissue
Abstract
Phenotypic characterization of cellular responses in equine infectious encephalitides has had limited description of both peripheral and resident cell populations in central nervous system (CNS) tissues due to limited species-specific reagents that react with formalin-fixed, paraffin embedded tissue (FFPE). This study identified a set of antibodies for investigating the immunopathology of infectious CNS diseases in horses. Multiple commercially available staining reagents and antibodies derived from antigens of various species for manual immunohistochemistry (IHC) were screened. Several techniques and reagents for heat-induced antigen retrieval, non-specific protein blocking, endogenous peroxidase blocking, and visualization-detection systems were tested during IHC protocol development. Boiling of slides in a low pH, citrate-based buffer solution in a double-boiler system was most consistent for epitope retrieval. Pressure-cooking, microwaving, high pH buffers, and proteinase K solutions often resulted in tissue disruption or no reactivity. Optimal blocking reagents and concentrations of each working antibody were determined. Ultimately, a set of monoclonal (mAb) and polyclonal antibodies (pAb) were identified for CD3(+) (pAb A0452, Dako) T-lymphocytes, CD79αcy(+) B-lymphocytes (mAb HM57, Dako), macrophages (mAb MAC387, Leica), NF-H(+) neurons (mAb NAP4, EnCor Biotechnology), microglia/macrophage (pAb Iba-1, Wako), and GFAP(+) astrocytes (mAb 5C10, EnCor Biotechnology). In paraffin embedded tissues, mAbs and pAbs derived from human and swine antigens were very successful at binding equine tissue targets. Individual, optimized protocols are provided for each positively reactive antibody for analyzing equine neuroinflammatory disease histopathology.
Keywords: Equine; Immunohistochemistry; Leukocytes; Neuroglia; Neuropathology.
Conflict of interest statement
The authors declare that they have no competing interests.
Figures
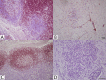
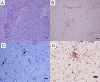


Similar articles
-
A method to generate antigen-specific mAb capable of staining formalin-fixed, paraffin-embedded tissue sections.J Immunol Methods. 2005 Apr;299(1-2):139-51. doi: 10.1016/j.jim.2005.02.006. Epub 2005 Apr 1. J Immunol Methods. 2005. PMID: 15896802
-
Evaluation of antibodies reactive with porcine lymphocytes and lymphoma cells in formalin-fixed, paraffin-embedded, antigen-retrieved tissue sections.Am J Vet Res. 1996 Jun;57(6):853-9. Am J Vet Res. 1996. PMID: 8725813
-
Immunohistochemical characterization of selected cell markers for the detection of hematopoietic cells in formalin-fixed, paraffin wax-embedded lymphoid tissues of harbor seals (Phoca vitulina) and walruses (Odobenus rosmarus rosmarus).Vet Immunol Immunopathol. 2010 Oct 15;137(3-4):305-9. doi: 10.1016/j.vetimm.2010.05.012. Epub 2010 Jun 2. Vet Immunol Immunopathol. 2010. PMID: 20566219
-
Immunohistochemical investigation of the cross-reactivity of selected cell markers from various species for characterization of lymphatic tissues in the harbour porpoise (Phocoena phocoena).J Comp Pathol. 2001 Nov;125(4):311-7. doi: 10.1053/jcpa.2001.0509. J Comp Pathol. 2001. PMID: 11798248
-
A Dermatopathologist's Guide to Troubleshooting Immunohistochemistry Part 1: Methods and Pitfalls.Am J Dermatopathol. 2015 Aug;37(8):593-603. doi: 10.1097/DAD.0000000000000335. Am J Dermatopathol. 2015. PMID: 26193707 Review.
Cited by
-
The seed oil of Paeonia ludlowii ameliorates Aβ25-35-induced Alzheimer's disease in rats.Food Sci Nutr. 2021 Mar 30;9(5):2402-2413. doi: 10.1002/fsn3.2102. eCollection 2021 May. Food Sci Nutr. 2021. PMID: 34026059 Free PMC article.
-
Direct interaction of HIV gp120 with neuronal CXCR4 and CCR5 receptors induces cofilin-actin rod pathology via a cellular prion protein- and NOX-dependent mechanism.PLoS One. 2021 Mar 11;16(3):e0248309. doi: 10.1371/journal.pone.0248309. eCollection 2021. PLoS One. 2021. PMID: 33705493 Free PMC article.
-
Naoling decoction restores cognitive function by inhibiting the neuroinflammatory network in a rat model of Alzheimer's disease.Oncotarget. 2017 Jun 27;8(26):42648-42663. doi: 10.18632/oncotarget.17337. Oncotarget. 2017. PMID: 28487495 Free PMC article.
-
Comparison of the clinical and radiographic appearance of the cervical vertebrae with histological and anatomical findings in an eight-month old warmblood stallion suffering from cervical vertebral stenotic myelopathy (CVSM).BMC Vet Res. 2019 Aug 15;15(1):296. doi: 10.1186/s12917-019-2047-x. BMC Vet Res. 2019. PMID: 31416466 Free PMC article.
-
Cellular distribution of cannabinoid-related receptors TRPV1, PPAR-gamma, GPR55 and GPR3 in the equine cervical dorsal root ganglia.Equine Vet J. 2021 Aug 21;54(4):788-98. doi: 10.1111/evj.13499. Online ahead of print. Equine Vet J. 2021. PMID: 34418142 Free PMC article.
References
-
- Boy MG, Galligan DT, Divers TJ. Protozoal encephalomyelitis in horses: 82 cases 1972–1986. Journal of the American Veterinary Medical Association. 1990;196(4):632–634. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

