Human monocytes undergo functional re-programming during differentiation to dendritic cell mediated by human extravillous trophoblasts
- PMID: 26857012
- PMCID: PMC4746586
- DOI: 10.1038/srep20409
Human monocytes undergo functional re-programming during differentiation to dendritic cell mediated by human extravillous trophoblasts
Abstract
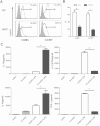
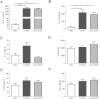
Maternal immune adaptation is required for a successful pregnancy to avoid rejection of the fetal-placental unit. Dendritic cells within the decidual microenvironment lock in a tolerogenic profile. However, how these tolerogenic DCs are induced and the underlying mechanisms are largely unknown. In this study, we show that human extravillous trophoblasts redirect the monocyte-to-DC transition and induce regulatory dendritic cells. DCs differentiated from blood monocytes in the presence of human extravillous trophoblast cell line HTR-8/SVneo displayed a DC-SIGN(+)CD14(+)CD1a(-) phenotype, similar with decidual DCs. HTR8-conditioned DCs were unable to develop a fully mature phenotype in response to LPS, and altered the cytokine secretory profile significantly. Functionally, conditioned DCs poorly induced the proliferation and activation of allogeneic T cells, whereas promoted CD4(+)CD25(+)Foxp3(+) Treg cells generation. Furthermore, the supernatant from DC and HTR-8/SVneo coculture system contained significant high amount of M-CSF and MCP-1. Using neutralizing antibodies, we discussed the role of M-CSF and MCP-1 during monocyte-to-DCs differentiation mediated by extravillous trophoblasts. Our data indicate that human extravillous trophoblasts play an important role in modulating the monocyte-to-DC differentiation through M-CSF and MCP-1, which facilitate the establishment of a tolerogenic microenvironment at the maternal-fetal interface.
Figures




Similar articles
-
Trophoblast cells induce a tolerogenic profile in dendritic cells.Hum Reprod. 2012 Sep;27(9):2598-606. doi: 10.1093/humrep/des208. Epub 2012 Jun 20. Hum Reprod. 2012. PMID: 22718280
-
MIC-1 (a multifunctional modulator of dendritic cell phenotype and function) is produced by decidual stromal cells and trophoblasts.Hum Reprod. 2012 Jan;27(1):200-9. doi: 10.1093/humrep/der358. Epub 2011 Nov 6. Hum Reprod. 2012. PMID: 22064648
-
DC-SIGN ligation greatly affects dendritic cell differentiation from monocytes compromising their normal function.J Leukoc Biol. 2011 Jun;89(6):893-905. doi: 10.1189/jlb.0810463. Epub 2011 Mar 29. J Leukoc Biol. 2011. PMID: 21447648
-
GM-CSF in the generation of dendritic cells from human blood monocyte precursors: recent advances.Immunobiology. 2008;213(9-10):859-70. doi: 10.1016/j.imbio.2008.07.017. Epub 2008 Sep 2. Immunobiology. 2008. PMID: 18926300 Review.
-
HLA-G Orchestrates the Early Interaction of Human Trophoblasts with the Maternal Niche.Front Immunol. 2015 Mar 30;6:128. doi: 10.3389/fimmu.2015.00128. eCollection 2015. Front Immunol. 2015. PMID: 25870595 Free PMC article. Review.
Cited by
-
Human Miscarriage Is Associated With Dysregulations in Peripheral Blood-Derived Myeloid Dendritic Cell Subsets.Front Immunol. 2019 Oct 15;10:2440. doi: 10.3389/fimmu.2019.02440. eCollection 2019. Front Immunol. 2019. PMID: 31681319 Free PMC article.
-
Human leukocyte antigen class II quantification by targeted mass spectrometry in dendritic-like cell lines and monocyte-derived dendritic cells.Sci Rep. 2021 Jan 13;11(1):1028. doi: 10.1038/s41598-020-77024-y. Sci Rep. 2021. PMID: 33441579 Free PMC article.
-
Human Decidual Stromal Cells as a Component of the Implantation Niche and a Modulator of Maternal Immunity.J Pregnancy. 2016;2016:8689436. doi: 10.1155/2016/8689436. Epub 2016 Apr 28. J Pregnancy. 2016. PMID: 27239344 Free PMC article. Review.
-
More Than Just Attractive: How CCL2 Influences Myeloid Cell Behavior Beyond Chemotaxis.Front Immunol. 2019 Dec 13;10:2759. doi: 10.3389/fimmu.2019.02759. eCollection 2019. Front Immunol. 2019. PMID: 31921102 Free PMC article. Review.
-
CCL2: An important cytokine in normal and pathological pregnancies: A review.Front Immunol. 2023 Jan 6;13:1053457. doi: 10.3389/fimmu.2022.1053457. eCollection 2022. Front Immunol. 2023. PMID: 36685497 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

