Robust Detection of DNA Hypermethylation of ZNF154 as a Pan-Cancer Locus with in Silico Modeling for Blood-Based Diagnostic Development
- PMID: 26857064
- PMCID: PMC4816708
- DOI: 10.1016/j.jmoldx.2015.11.004
Robust Detection of DNA Hypermethylation of ZNF154 as a Pan-Cancer Locus with in Silico Modeling for Blood-Based Diagnostic Development
Abstract
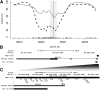
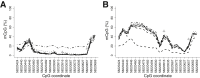
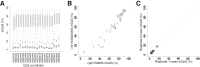
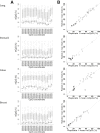
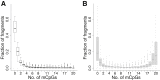
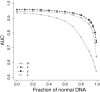
Sites that display recurrent, aberrant DNA methylation in cancer represent potential biomarkers for screening and diagnostics. Previously, we identified hypermethylation at the ZNF154 CpG island in 15 solid epithelial tumor types from 13 different organs. In this study, we measure the magnitude and pattern of differential methylation of this region across colon, lung, breast, stomach, and endometrial tumor samples using next-generation bisulfite amplicon sequencing. We found that all tumor types and subtypes are hypermethylated at this locus compared with normal tissue. To evaluate this site as a possible pan-cancer marker, we compare the ability of several sequence analysis methods to distinguish the five tumor types (184 tumor samples) from normal tissue samples (n = 34). The classification performance for the strongest method, measured by the area under (the receiver operating characteristic) curve (AUC), is 0.96, close to a perfect value of 1. Furthermore, in a computational simulation of circulating tumor DNA, we were able to detect limited amounts of tumor DNA diluted with normal DNA: 1% tumor DNA in 99% normal DNA yields AUCs of up to 0.79. Our findings suggest that hypermethylation of the ZNF154 CpG island is a relevant biomarker for identifying solid tumor DNA and may have utility as a generalizable biomarker for circulating tumor DNA.
Published by Elsevier Inc.
Figures



References
-
- Siegel R., Ma J., Zou Z., Jemal A. Cancer statistics, 2014. CA Cancer J Clin. 2014;64:9–29. - PubMed
-
- Edwards B.K., Noone A.M., Mariotto A.B., Simard E.P., Boscoe F.P., Henley S.J., Jemal A., Cho H., Anderson R.N., Kohler B.A., Eheman C.R., Ward E.M. Annual Report to the Nation on the status of cancer, 1975-2010, featuring prevalence of comorbidity and impact on survival among persons with lung, colorectal, breast, or prostate cancer. Cancer. 2014;120:1290–1314. - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

