All-optical control of cardiac excitation: combined high-resolution optogenetic actuation and optical mapping
- PMID: 26857427
- PMCID: PMC4850200
- DOI: 10.1113/JP271559
All-optical control of cardiac excitation: combined high-resolution optogenetic actuation and optical mapping
Abstract
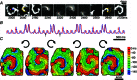
Cardiac tissue is an excitable system that can support complex spatiotemporal dynamics, including instabilities (arrhythmias) with lethal consequences. While over the last two decades optical mapping of excitation (voltage and calcium dynamics) has facilitated the detailed characterization of such arrhythmia events, until recently, no precise tools existed to actively interrogate cardiac dynamics in space and time. In this work, we discuss the combined use of new methods for space- and time-resolved optogenetic actuation and simultaneous fast, high resolution optical imaging of cardiac excitation waves. First, the mechanisms, limitations and unique features of optically induced responses in cardiomyocytes are outlined. These include the ability to bidirectionally control the membrane potential using depolarizing and hyperpolarizing opsins; the ability to induce prolonged sustained voltage changes; and the ability to control refractoriness and the shape of the cardiac action potential. At the syncytial tissue level, we discuss optogenetically enabled experimentation on cell-cell coupling, alteration of conduction properties and termination of propagating waves by light. Specific attention is given to space- and time-resolved application of optical stimulation using dynamic light patterns to perturb ongoing activation and to probe electrophysiological properties at desired tissue locations. The combined use of optical methods to perturb and to observe the system can offer new tools for precise feedback control of cardiac electrical activity, not available previously with pharmacological and electrical stimulation. These new experimental tools for all-optical electrophysiology allow for a level of precise manipulation and quantification of cardiac dynamics comparable in robustness to the computational setting, and can provide new insights into pacemaking, arrhythmogenesis and suppression or cardioversion.
© 2016 The Authors. The Journal of Physiology published by John Wiley & Sons Ltd on behalf of The Physiological Society.
Figures
Similar articles
-
Principles of Optogenetic Methods and Their Application to Cardiac Experimental Systems.Front Physiol. 2019 Sep 11;10:1096. doi: 10.3389/fphys.2019.01096. eCollection 2019. Front Physiol. 2019. PMID: 31572204 Free PMC article. Review.
-
Cardiac Optogenetics and Optical Mapping - Overcoming Spectral Congestion in All-Optical Cardiac Electrophysiology.Front Physiol. 2019 Mar 7;10:182. doi: 10.3389/fphys.2019.00182. eCollection 2019. Front Physiol. 2019. PMID: 30899227 Free PMC article. Review.
-
Real-time optical manipulation of cardiac conduction in intact hearts.J Physiol. 2018 Sep;596(17):3841-3858. doi: 10.1113/JP276283. Epub 2018 Aug 7. J Physiol. 2018. PMID: 29989169 Free PMC article.
-
Optical mapping and optogenetics in cardiac electrophysiology research and therapy: a state-of-the-art review.Europace. 2024 Feb 1;26(2):euae017. doi: 10.1093/europace/euae017. Europace. 2024. PMID: 38227822 Free PMC article. Review.
-
Cardiac applications of optogenetics.Prog Biophys Mol Biol. 2014 Aug;115(2-3):294-304. doi: 10.1016/j.pbiomolbio.2014.07.001. Epub 2014 Jul 15. Prog Biophys Mol Biol. 2014. PMID: 25035999 Free PMC article. Review.
Cited by
-
Anion channelrhodopsins for inhibitory cardiac optogenetics.Sci Rep. 2016 Sep 15;6:33530. doi: 10.1038/srep33530. Sci Rep. 2016. PMID: 27628215 Free PMC article.
-
Optogenetics: Background, Methodological Advances and Potential Applications for Cardiovascular Research and Medicine.Front Bioeng Biotechnol. 2020 Jan 29;7:466. doi: 10.3389/fbioe.2019.00466. eCollection 2019. Front Bioeng Biotechnol. 2020. PMID: 32064254 Free PMC article. Review.
-
A flexible and versatile system for multi-color fiber photometry and optogenetic manipulation.Cell Rep Methods. 2023 Mar 7;3(3):100418. doi: 10.1016/j.crmeth.2023.100418. eCollection 2023 Mar 27. Cell Rep Methods. 2023. PMID: 37056369 Free PMC article.
-
Light-Activated Dynamic Clamp Using iPSC-Derived Cardiomyocytes.Biophys J. 2018 Dec 4;115(11):2206-2217. doi: 10.1016/j.bpj.2018.10.018. Epub 2018 Oct 30. Biophys J. 2018. PMID: 30447994 Free PMC article.
-
Principles of Optogenetic Methods and Their Application to Cardiac Experimental Systems.Front Physiol. 2019 Sep 11;10:1096. doi: 10.3389/fphys.2019.01096. eCollection 2019. Front Physiol. 2019. PMID: 31572204 Free PMC article. Review.
References
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

