The fungal-specific transcription factor Vdpf influences conidia production, melanized microsclerotia formation and pathogenicity in Verticillium dahliae
- PMID: 26857810
- PMCID: PMC6638448
- DOI: 10.1111/mpp.12367
The fungal-specific transcription factor Vdpf influences conidia production, melanized microsclerotia formation and pathogenicity in Verticillium dahliae
Abstract
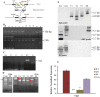
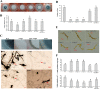
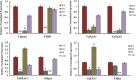
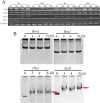
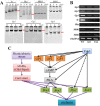
Verticillium dahliae is a soil-borne, hemibiotrophic phytopathogenic fungus that causes wilting in crop plants. Here, we constructed a random insertional mutant library using Agrobacterium tumefaciens-mediated transformation to study the pathogenicity and regulatory mechanisms of V. dahliae. The fungal-specific transcription factor-encoding gene Vdpf was shown to be associated with vegetative growth and virulence, with the highest transcript expression occurring during conidia formation in the V991 strain. The deletion mutants (ΔVdpf) and insertion mutants (IMΔVdpf) produced fewer conidia than did the wild-type (WT) fungi, which contributed to the reduced virulence. Unlike the WT, the complemented strains and IMΔVdpf, ΔVdpf formed swollen, thick-walled and hyaline mycelium rather than melanized microsclerotia. The ΔVdpf mutants were melanin deficient, with undetectable expression of melanin biosynthesis-related genes (Brn1, Brn2 and Scd1). The melanin deficiency was related to cyclic adenosine monophosphate (cAMP) and the G-protein-coupled signalling pathways in this study. Similar to the WT and complemented strains, the ΔVdpf and IMΔVdpf mutants could also successfully penetrate into cotton and tobacco roots, but displayed reduced virulence because of lower biomass in the plant roots and significantly reduced expression of pathogenicity-related genes in V. dahliae. In conclusion, these results provide insights into the role of Vdpf in melanized microsclerotia formation, conidia production and pathogenicity.
Keywords: Verticillium dahliae; conidiation; fungal-specific transcription factor; melanized microsclerotia; pathogenicity.
© 2016 BSPP and John Wiley & Sons Ltd.
Figures


Similar articles
-
The bZip Transcription Factor VdMRTF1 is a Negative Regulator of Melanin Biosynthesis and Virulence in Verticillium dahliae.Microbiol Spectr. 2022 Apr 27;10(2):e0258121. doi: 10.1128/spectrum.02581-21. Epub 2022 Apr 11. Microbiol Spectr. 2022. PMID: 35404080 Free PMC article.
-
Identification of pathogenicity-related genes in the vascular wilt fungus Verticillium dahliae by Agrobacterium tumefaciens-mediated T-DNA insertional mutagenesis.Mol Biotechnol. 2011 Nov;49(3):209-21. doi: 10.1007/s12033-011-9392-8. Mol Biotechnol. 2011. PMID: 21424547 Free PMC article.
-
VdPKS1 is required for melanin formation and virulence in a cotton wilt pathogen Verticillium dahliae.Sci China Life Sci. 2017 Aug;60(8):868-879. doi: 10.1007/s11427-017-9075-3. Epub 2017 Jul 26. Sci China Life Sci. 2017. PMID: 28755294
-
Interactions between Verticillium dahliae and its host: vegetative growth, pathogenicity, plant immunity.Appl Microbiol Biotechnol. 2014 Aug;98(16):6921-32. doi: 10.1007/s00253-014-5863-8. Epub 2014 Jun 15. Appl Microbiol Biotechnol. 2014. PMID: 24928658 Review.
-
Key Insights and Research Prospects at the Dawn of the Population Genomics Era for Verticillium dahliae.Annu Rev Phytopathol. 2021 Aug 25;59:31-51. doi: 10.1146/annurev-phyto-020620-121925. Epub 2021 Apr 23. Annu Rev Phytopathol. 2021. PMID: 33891830 Review.
Cited by
-
The Kelch Repeat Protein VdKeR1 Is Essential for Development, Ergosterol Metabolism, and Virulence in Verticillium dahliae.J Fungi (Basel). 2024 Sep 9;10(9):643. doi: 10.3390/jof10090643. J Fungi (Basel). 2024. PMID: 39330403 Free PMC article.
-
Identification of VdASP F2-interacting protein as a regulator of microsclerotial formation in Verticillium dahliae.Microb Biotechnol. 2022 Jul;15(7):2040-2054. doi: 10.1111/1751-7915.14066. Epub 2022 Apr 27. Microb Biotechnol. 2022. PMID: 35478269 Free PMC article.
-
cAMP Signalling Pathway in Biocontrol Fungi.Curr Issues Mol Biol. 2022 Jun 4;44(6):2622-2634. doi: 10.3390/cimb44060179. Curr Issues Mol Biol. 2022. PMID: 35735620 Free PMC article. Review.
-
The Frq-Frh Complex Light-Dependently Delays Sfl1-Induced Microsclerotia Formation in Verticillium dahliae.J Fungi (Basel). 2023 Jul 4;9(7):725. doi: 10.3390/jof9070725. J Fungi (Basel). 2023. PMID: 37504714 Free PMC article.
-
Target of rapamycin controls hyphal growth and pathogenicity through FoTIP4 in Fusarium oxysporum.Mol Plant Pathol. 2021 Oct;22(10):1239-1255. doi: 10.1111/mpp.13108. Epub 2021 Jul 20. Mol Plant Pathol. 2021. PMID: 34288333 Free PMC article.
References
-
- Badis, G. , Chan, E.T. , van Bakel, H. , Pena‐Castillo, L. , Tillo, D. , Tsui, K. , Carlson, C.D. , Gossett, A.J. , Hasinoff, M.J. , Warren, C.L. , Gebbia, M. , Talukder, S. , Yang, A. , Mnaimneh, S. , Terterov, D. , Coburn, D. , Li Yeo, A. , Yeo, Z.X. , Clarke, N.D. , Lieb, J.D. , Ansari, A.Z. , Nislow, C. and Hughes, T.R. (2008) A library of yeast transcription factor motifs reveals a widespread function for Rsc3 in targeting nucleosome exclusion at promoters. Molecular Cell, 32, 878–887. - PMC - PubMed
-
- Bell, A. (2001) Stem and root diseases, Verticillium wilt In: Compendium of Cotton Disease (Kirkpatrick T.L. and Rothrock C.S., eds), pp. 28–31. St. Paul, MN: The American Phytopathological Society.
-
- Bell, A.A. and Wheeler, M.H. (1986) Biosynthesis and functions of fungal melanins. Annu. Rev. Phytopathol. 24, 411–451.
-
- Bell, A.A. , Puhalla, J.E. , Tolmsoff, W.J. and Stipanovic, R.D. (1976) Use of mutants to establish (+)‐scytalone as an intermediate in melanin biosynthesis by Verticillium dahliae . Can. J. Microbiol. 22, 787–799. - PubMed
-
- Bhat, R.G. , Smith, R.F. , Koike, S.T. , Wu, B.M. and Subbarao, K.V. (2003) Characterization of Verticillium dahliae isolates and wilt epidemics of pepper. Plant Dis. 87, 789–797. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

