Inhibition of the Formation of the Spf1p Phosphoenzyme by Ca2
- PMID: 26858246
- PMCID: PMC4817200
- DOI: 10.1074/jbc.M115.695122
Inhibition of the Formation of the Spf1p Phosphoenzyme by Ca2
Abstract
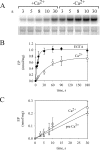
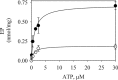
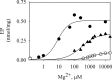
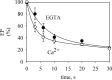
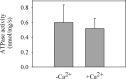
P5-ATPases are important for processes associated with the endosomal-lysosomal system of eukaryotic cells. In humans, the loss of function of P5-ATPases causes neurodegeneration. In the yeastSaccharomyces cerevisiae, deletion of P5-ATPase Spf1p gives rise to endoplasmic reticulum stress. The reaction cycle of P5-ATPases is poorly characterized. Here, we showed that the formation of the Spf1p catalytic phosphoenzyme was fast in a reaction medium containing ATP, Mg(2+), and EGTA. Low concentrations of Ca(2+)in the phosphorylation medium decreased the rate of phosphorylation and the maximal level of phosphoenzyme. Neither Mn(2+)nor Mg(2+)had an inhibitory effect on the formation of the phosphoenzyme similar to that of Ca(2+) TheKmfor ATP in the phosphorylation reaction was ∼1 μmand did not significantly change in the presence of Ca(2+) Half-maximal phosphorylation was attained at 8 μmMg(2+), but higher concentrations partially protected from Ca(2+)inhibition. In conditions similar to those used for phosphorylation, Ca(2+)had a small effect accelerating dephosphorylation and minimally affected ATPase activity, suggesting that the formation of the phosphoenzyme was not the limiting step of the ATP hydrolytic cycle.
Keywords: ATPase; P-ATPase; P5-ATPase; ion pump; phosphoryl transfer; phosphorylation; protein phosphorylation; transporter.
© 2016 by The American Society for Biochemistry and Molecular Biology, Inc.
Figures


References
-
- Palmgren M. G., Axelsen K. B. (1998) Evolution of P-type ATPases. Biochim. Biophys. Acta 1365, 37–45 - PubMed
-
- Sørensen D. M., Buch-Pedersen M. J., and Palmgren M. G. (2010) Structural divergence between the two subgroups of P5 ATPases. Biochim. Biophys. Acta 1797, 846–855 - PubMed
-
- Perrett R. M., Alexopoulou Z., and Tofaris G. K. (2015) The endosomal pathway in Parkinson's disease. Mol. Cell Neurosci. 66, 21–28 - PubMed
-
- Suzuki C., and Shimma Y. I. (1999) P-type ATPase Spf1 mutants show a novel resistance mechanism for the killer toxin SMKT. Mol. Microbiol. 32, 813–823 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

