Functional Imaging of Autonomic Regulation: Methods and Key Findings
- PMID: 26858595
- PMCID: PMC4726771
- DOI: 10.3389/fnins.2015.00513
Functional Imaging of Autonomic Regulation: Methods and Key Findings
Abstract
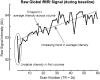
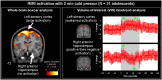
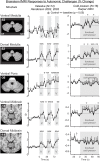
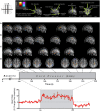
Central nervous system processing of autonomic function involves a network of regions throughout the brain which can be visualized and measured with neuroimaging techniques, notably functional magnetic resonance imaging (fMRI). The development of fMRI procedures has both confirmed and extended earlier findings from animal models, and human stroke and lesion studies. Assessments with fMRI can elucidate interactions between different central sites in regulating normal autonomic patterning, and demonstrate how disturbed systems can interact to produce aberrant regulation during autonomic challenges. Understanding autonomic dysfunction in various illnesses reveals mechanisms that potentially lead to interventions in the impairments. The objectives here are to: (1) describe the fMRI neuroimaging methodology for assessment of autonomic neural control, (2) outline the widespread, lateralized distribution of function in autonomic sites in the normal brain which includes structures from the neocortex through the medulla and cerebellum, (3) illustrate the importance of the time course of neural changes when coordinating responses, and how those patterns are impacted in conditions of sleep-disordered breathing, and (4) highlight opportunities for future research studies with emerging methodologies. Methodological considerations specific to autonomic testing include timing of challenges relative to the underlying fMRI signal, spatial resolution sufficient to identify autonomic brainstem nuclei, blood pressure, and blood oxygenation influences on the fMRI signal, and the sustained timing, often measured in minutes of challenge periods and recovery. Key findings include the lateralized nature of autonomic organization, which is reminiscent of asymmetric motor, sensory, and language pathways. Testing brain function during autonomic challenges demonstrate closely-integrated timing of responses in connected brain areas during autonomic challenges, and the involvement with brain regions mediating postural and motoric actions, including respiration, and cardiac output. The study of pathological processes associated with autonomic disruption shows susceptibilities of different brain structures to altered timing of neural function, notably in sleep disordered breathing, such as obstructive sleep apnea and congenital central hypoventilation syndrome. The cerebellum, in particular, serves coordination roles for vestibular stimuli and blood pressure changes, and shows both injury and substantially altered timing of responses to pressor challenges in sleep-disordered breathing conditions. The insights into central autonomic processing provided by neuroimaging have assisted understanding of such regulation, and may lead to new treatment options for conditions with disrupted autonomic function.
Keywords: SIDS; SUDEP; cerebellum; fMRI; insula; parasympathetic; sleep-disordered breathing; sympathetic.
Figures

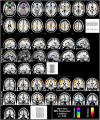
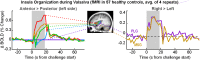
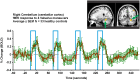
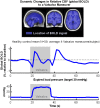
Similar articles
-
Functional neuroanatomy and sleep-disordered breathing: implications for autonomic regulation.Anat Rec (Hoboken). 2012 Sep;295(9):1385-95. doi: 10.1002/ar.22514. Epub 2012 Jul 31. Anat Rec (Hoboken). 2012. PMID: 22851218 Review.
-
Central autonomic regulation in congenital central hypoventilation syndrome.Neuroscience. 2010 Jun 2;167(4):1249-56. doi: 10.1016/j.neuroscience.2010.02.078. Epub 2010 Mar 6. Neuroscience. 2010. PMID: 20211704 Free PMC article.
-
Sleep-disordered breathing: effects on brain structure and function.Respir Physiol Neurobiol. 2013 Sep 15;188(3):383-91. doi: 10.1016/j.resp.2013.04.021. Epub 2013 May 1. Respir Physiol Neurobiol. 2013. PMID: 23643610 Free PMC article. Review.
-
Impaired cerebellar and limbic responses to the valsalva maneuver in heart failure.Cerebellum. 2012 Dec;11(4):931-8. doi: 10.1007/s12311-012-0361-y. Cerebellum. 2012. PMID: 22370874
-
Visualization of sleep influences on cerebellar and brainstem cardiac and respiratory control mechanisms.Brain Res Bull. 2000 Sep 1;53(1):125-31. doi: 10.1016/s0361-9230(00)00317-8. Brain Res Bull. 2000. PMID: 11033217 Review.
Cited by
-
Imaging fascicular organization of rat sciatic nerves with fast neural electrical impedance tomography.Nat Commun. 2020 Dec 7;11(1):6241. doi: 10.1038/s41467-020-20127-x. Nat Commun. 2020. PMID: 33288760 Free PMC article.
-
Altered Relationship Between Heart Rate Variability and fMRI-Based Functional Connectivity in People With Epilepsy.Front Neurol. 2021 Jun 10;12:671890. doi: 10.3389/fneur.2021.671890. eCollection 2021. Front Neurol. 2021. PMID: 34177777 Free PMC article.
-
Novel indolyl-chalcone derivatives inhibit A549 lung cancer cell growth through activating Nrf-2/HO-1 and inducing apoptosis in vitro and in vivo.Sci Rep. 2017 Jun 20;7(1):3919. doi: 10.1038/s41598-017-04411-3. Sci Rep. 2017. PMID: 28634389 Free PMC article.
-
Precision Medicine for Breath-Focused Mind-Body Therapies for Stress and Anxiety: Are We Ready Yet?Glob Adv Health Med. 2021 Jan 13;10:2164956120986129. doi: 10.1177/2164956120986129. eCollection 2021. Glob Adv Health Med. 2021. PMID: 33489480 Free PMC article.
-
Dysfunctional Brain Networking among Autonomic Regulatory Structures in Temporal Lobe Epilepsy Patients at High Risk of Sudden Unexpected Death in Epilepsy.Front Neurol. 2017 Oct 16;8:544. doi: 10.3389/fneur.2017.00544. eCollection 2017. Front Neurol. 2017. PMID: 29085330 Free PMC article.
References
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

