Bicarbonate and Ca(2+) Sensing Modulators Activate Photoreceptor ROS-GC1 Synergistically
- PMID: 26858600
- PMCID: PMC4729890
- DOI: 10.3389/fnmol.2016.00005
Bicarbonate and Ca(2+) Sensing Modulators Activate Photoreceptor ROS-GC1 Synergistically
Abstract
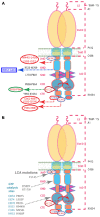
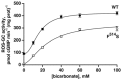
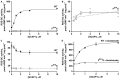
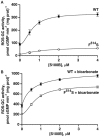
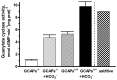
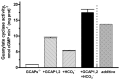
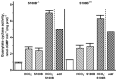
Photoreceptor ROS-GC1, a prototype subfamily member of the membrane guanylate cyclase family, is a central component of phototransduction. It is a single transmembrane-spanning protein, composed of modular blocks. In rods, guanylate cyclase activating proteins (GCAPs) 1 and 2 bind to its juxtamembrane domain (JMD) and the C-terminal extension, respectively, to accelerate cyclic GMP synthesis when Ca(2+) levels are low. In cones, the additional expression of the Ca(2+)-dependent guanylate cyclase activating protein (CD-GCAP) S100B which binds to its C-terminal extension, supports acceleration of cyclic GMP synthesis at high Ca(2+) levels. Independent of Ca(2+), ROS-GC1 activity is also stimulated directly by bicarbonate binding to the core catalytic domain (CCD). Several enticing molecular features of this transduction system are revealed in the present study. In combination, bicarbonate and Ca(2+)-dependent modulators raised maximal ROS-GC activity to levels that exceeded the sum of their individual effects. The F(514)S mutation in ROS-GC1 that causes blindness in type 1 Leber's congenital amaurosis (LCA) severely reduced basal ROS-GC1 activity. GCAP2 and S100B Ca(2+) signaling modes remained functional, while the GCAP1-modulated mode was diminished. Bicarbonate nearly restored basal activity as well as GCAP2- and S100B-stimulated activities of the F(514)S mutant to normal levels but could not resurrect GCAP1 stimulation. We conclude that GCAP1 and GCAP2 forge distinct pathways through domain-specific modules of ROS-GC1 whereas the S100B and GCAP2 pathways may overlap. The synergistic interlinking of bicarbonate to GCAPs- and S100B-modulated pathways intensifies and tunes the dependence of cyclic GMP synthesis on intracellular Ca(2+). Our study challenges the recently proposed GCAP1 and GCAP2 "overlapping" phototransduction model (Peshenko et al., 2015b).
Keywords: S100 proteins; bicarbonate; calcium-binding protein; guanylate cyclase (guanylyl cyclase); guanylate cyclase activating protein (GCAP); phototransduction; second messenger; signal transduction.
Figures

Similar articles
-
A second calcium regulator of rod outer segment membrane guanylate cyclase, ROS-GC1: neurocalcin.Biochemistry. 1999 Sep 28;38(39):12614-20. doi: 10.1021/bi990851n. Biochemistry. 1999. PMID: 10504230
-
Differential Ca(2+) sensor guanylate cyclase activating protein modes of photoreceptor rod outer segment membrane guanylate cyclase signaling.Biochemistry. 2012 Jun 12;51(23):4650-7. doi: 10.1021/bi300572w. Epub 2012 Jun 1. Biochemistry. 2012. PMID: 22642846 Free PMC article.
-
Impairment of the rod outer segment membrane guanylate cyclase dimerization in a cone-rod dystrophy results in defective calcium signaling.Biochemistry. 2000 Oct 17;39(41):12522-33. doi: 10.1021/bi001514d. Biochemistry. 2000. PMID: 11027131
-
Membrane guanylate cyclase, a multimodal transduction machine: history, present, and future directions.Front Mol Neurosci. 2014 Jul 2;7:56. doi: 10.3389/fnmol.2014.00056. eCollection 2014. Front Mol Neurosci. 2014. PMID: 25071437 Free PMC article. Review.
-
ROS-GC interlocked Ca(2+)-sensor S100B protein signaling in cone photoreceptors: review.Front Mol Neurosci. 2014 Mar 26;7:21. doi: 10.3389/fnmol.2014.00021. eCollection 2014. Front Mol Neurosci. 2014. PMID: 24723847 Free PMC article. Review.
Cited by
-
Calcium-sensor proteins but not bicarbonate ion activate retinal photoreceptor membrane guanylyl cyclase in photoreceptors.Front Mol Neurosci. 2024 Dec 9;17:1509366. doi: 10.3389/fnmol.2024.1509366. eCollection 2024. Front Mol Neurosci. 2024. PMID: 39717564 Free PMC article.
-
Molecular Details of Retinal Guanylyl Cyclase 1/GCAP-2 Interaction.Front Mol Neurosci. 2018 Sep 19;11:330. doi: 10.3389/fnmol.2018.00330. eCollection 2018. Front Mol Neurosci. 2018. PMID: 30283299 Free PMC article.
-
Membrane Guanylate Cyclase catalytic Subdomain: Structure and Linkage with Calcium Sensors and Bicarbonate.Front Mol Neurosci. 2017 Jun 7;10:173. doi: 10.3389/fnmol.2017.00173. eCollection 2017. Front Mol Neurosci. 2017. PMID: 28638321 Free PMC article.
-
CO2/bicarbonate modulates cone photoreceptor ROS-GC1 and restores its CORD6-linked catalytic activity.Mol Cell Biochem. 2018 Nov;448(1-2):91-105. doi: 10.1007/s11010-018-3317-9. Epub 2018 Feb 9. Mol Cell Biochem. 2018. PMID: 29427171 Free PMC article.
-
Effects of cell size and bicarbonate on single photon response variability in retinal rods.Front Mol Neurosci. 2022 Dec 14;15:1050545. doi: 10.3389/fnmol.2022.1050545. eCollection 2022. Front Mol Neurosci. 2022. PMID: 36590910 Free PMC article.
References
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

