Prenatal Choline Supplementation Diminishes Early-Life Iron Deficiency-Induced Reprogramming of Molecular Networks Associated with Behavioral Abnormalities in the Adult Rat Hippocampus
- PMID: 26865644
- PMCID: PMC4763487
- DOI: 10.3945/jn.115.227561
Prenatal Choline Supplementation Diminishes Early-Life Iron Deficiency-Induced Reprogramming of Molecular Networks Associated with Behavioral Abnormalities in the Adult Rat Hippocampus
Abstract
Background: Early-life iron deficiency is a common nutrient deficiency worldwide. Maternal iron deficiency increases the risk of schizophrenia and autism in the offspring. Postnatal iron deficiency in young children results in cognitive and socioemotional abnormalities in adulthood despite iron treatment. The rat model of diet-induced fetal-neonatal iron deficiency recapitulates the observed neurobehavioral deficits.
Objectives: We sought to establish molecular underpinnings for the persistent psychopathologic effects of early-life iron deficiency by determining whether it permanently reprograms the hippocampal transcriptome. We also assessed the effects of maternal dietary choline supplementation on the offspring's hippocampal transcriptome to identify pathways through which choline mitigates the emergence of long-term cognitive deficits.
Methods: Male rat pups were made iron deficient (ID) by providing pregnant and nursing dams an ID diet (4 g Fe/kg) from gestational day (G) 2 through postnatal day (PND) 7 and an iron-sufficient (IS) diet (200 g Fe/kg) thereafter. Control pups were provided IS diet throughout. Choline (5 g/kg) was given to half the pregnant dams in each group from G11 to G18. PND65 hippocampal transcriptomes were assayed by next generation sequencing (NGS) and analyzed with the use of knowledge-based Ingenuity Pathway Analysis. Real-time polymerase chain reaction was performed to validate a subset of altered genes.
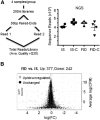
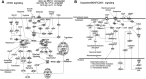
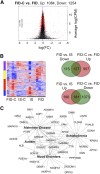
Results: Formerly ID rats had altered hippocampal expression of 619 from >10,000 gene loci sequenced by NGS, many of which map onto molecular networks implicated in psychological disorders, including anxiety, autism, and schizophrenia. There were significant interactions between iron status and prenatal choline treatment in influencing gene expression. Choline supplementation reduced the effects of iron deficiency, including those on gene networks associated with autism and schizophrenia.
Conclusions: Fetal-neonatal iron deficiency reprograms molecular networks associated with the pathogenesis of neurologic and psychological disorders in adult rats. The positive response to prenatal choline represents a potential adjunctive therapeutic supplement to the high-risk group.
Keywords: choline supplementation; fetal iron deficiency; hippocampus; psychological disorders; transcriptome.
© 2016 American Society for Nutrition.
Conflict of interest statement
Author disclosures: PV Tran, BC Kennedy, MT Pisansky, K-J Won, JC Gewirtz, RA Simmons, and MK Georgieff, no conflicts of interest.
Figures


Similar articles
-
Beneficial effects of postnatal choline supplementation on long-Term neurocognitive deficit resulting from fetal-Neonatal iron deficiency.Behav Brain Res. 2018 Jan 15;336:40-43. doi: 10.1016/j.bbr.2017.07.043. Epub 2017 Aug 12. Behav Brain Res. 2018. PMID: 28811181 Free PMC article.
-
Prenatal choline supplementation ameliorates the long-term neurobehavioral effects of fetal-neonatal iron deficiency in rats.J Nutr. 2014 Nov;144(11):1858-65. doi: 10.3945/jn.114.198739. Epub 2014 Sep 3. J Nutr. 2014. PMID: 25332485 Free PMC article.
-
Identification of Genes Responding to Iron or Choline Treatment for Early-Life Iron Deficiency in the Male Rat Hippocampal Transcriptomes.J Nutr. 2024 Apr;154(4):1141-1152. doi: 10.1016/j.tjnut.2024.02.021. Epub 2024 Feb 24. J Nutr. 2024. PMID: 38408730 Free PMC article.
-
The fetal origins of memory: the role of dietary choline in optimal brain development.J Pediatr. 2006 Nov;149(5 Suppl):S131-6. doi: 10.1016/j.jpeds.2006.06.065. J Pediatr. 2006. PMID: 17212955 Free PMC article. Review.
-
The role of iron in neurodevelopment: fetal iron deficiency and the developing hippocampus.Biochem Soc Trans. 2008 Dec;36(Pt 6):1267-71. doi: 10.1042/BST0361267. Biochem Soc Trans. 2008. PMID: 19021538 Free PMC article. Review.
Cited by
-
The dynamic inflammatory profile of pregnancy can be monitored using a novel lipid-based mass spectrometry technique.Mol Omics. 2023 May 9;19(4):340-350. doi: 10.1039/d2mo00294a. Mol Omics. 2023. PMID: 36883215 Free PMC article.
-
Atypical fetal development: Fetal alcohol syndrome, nutritional deprivation, teratogens, and risk for neurodevelopmental disorders and psychopathology.Dev Psychopathol. 2018 Aug;30(3):1063-1086. doi: 10.1017/S0954579418000500. Dev Psychopathol. 2018. PMID: 30068419 Free PMC article. Review.
-
Prenatal Folate and Choline Levels and Brain and Cognitive Development in Children: A Critical Narrative Review.Nutrients. 2022 Jan 15;14(2):364. doi: 10.3390/nu14020364. Nutrients. 2022. PMID: 35057545 Free PMC article. Review.
-
Early-Life Neuronal-Specific Iron Deficiency Alters the Adult Mouse Hippocampal Transcriptome.J Nutr. 2018 Oct 1;148(10):1521-1528. doi: 10.1093/jn/nxy125. J Nutr. 2018. PMID: 30169712 Free PMC article.
-
The Impact of Traditional Food and Lifestyle Behavior on Epigenetic Burden of Chronic Disease.Glob Chall. 2017 Oct 27;1(8):1700043. doi: 10.1002/gch2.201700043. eCollection 2017 Nov 16. Glob Chall. 2017. PMID: 31565292 Free PMC article. Review.
References
-
- Georgieff MK, Wewerka SW, Nelson CA, Deregnier RA. Iron status at 9 months of infants with low iron stores at birth. J Pediatr 2002;141:405–9. - PubMed
-
- Petry CD, Eaton MA, Wobken JD, Mills MM, Johnson DE, Georgieff MK. Iron deficiency of liver, heart, and brain in newborn infants of diabetic mothers. J Pediatr 1992;121:109–14. - PubMed
-
- Chockalingam UM, Murphy E, Ophoven JC, Weisdorf SA, Georgieff MK. Cord transferrin and ferritin values in newborn infants at risk for prenatal uteroplacental insufficiency and chronic hypoxia. J Pediatr 1987;111:283–6. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

