The Low-pH Resistance of Neuraminidase Is Essential for the Replication of Influenza A Virus in Duck Intestine following Infection via the Oral Route
- PMID: 26865722
- PMCID: PMC4810549
- DOI: 10.1128/JVI.03107-15
The Low-pH Resistance of Neuraminidase Is Essential for the Replication of Influenza A Virus in Duck Intestine following Infection via the Oral Route
Abstract
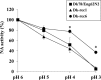
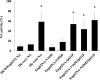
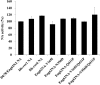
Influenza A viruses are known to primarily replicate in duck intestine following infection via the oral route, but the specific role of neuraminidase (NA) for the intestinal tropism of influenza A viruses has been unclear. A reassortant virus (Dk78/Eng62N2) did not propagate in ducks infected via the oral route. To generate variant viruses that grow well in ducks via the oral route, we isolated viruses that effectively replicate in intestinal mucosal cells by passaging Dk78/Eng62N2 in duck via rectal-route infection. This procedure led to the isolation of a variant virus from the duck intestine. This virus was propagated using embryonated chicken eggs and inoculated into a duck via the oral route, which led to the isolation of Dk-rec6 from the duck intestine. Experimental infections with mutant viruses generated by using reverse genetics indicated that the paired mutation of residues 356 and 431 in NA was necessary for the viral replication in duck intestine. The NA assay revealed that the activity of Dk78/Eng62N2 almost disappeared after pH 3 treatment, whereas that of Dk-rec6 was maintained. Furthermore, to identify the amino acid residues associated with the low-pH resistance, we measured the activities of mutant NA proteins transiently expressed in 293 cells after pH 3 treatment. All mutant NA proteins that possessed proline at position 431 showed higher activities than NA proteins that possessed glutamine at this position. These findings indicate that the low-pH resistance of NA plays an important role in the ability of influenza A virus to replicate in duck intestine.
Importance: Neuraminidase (NA) activity facilitates the release of viruses from cells and, as such, is important for the replicative efficiency of influenza A virus. Ducks are believed to serve as the principal natural reservoir for influenza A virus; however, the key properties of NA for viral infection in duck are not well understood. In this study, we identify amino acid residues in NA that contribute to viral replication in ducks via the natural route of infection and demonstrate that maintenance of NA activity under low-pH conditions is associated with the biological properties of the virus. These findings provide insights into the mechanisms of replication of influenza A virus in ducks.
Copyright © 2016, American Society for Microbiology. All Rights Reserved.
Figures
Similar articles
-
Amino acids responsible for the absolute sialidase activity of the influenza A virus neuraminidase: relationship to growth in the duck intestine.J Virol. 2001 Dec;75(23):11773-80. doi: 10.1128/JVI.75.23.11773-11780.2001. J Virol. 2001. PMID: 11689658 Free PMC article.
-
Human influenza a viral genes responsible for the restriction of its replication in duck intestine.Virology. 2002 Apr 10;295(2):250-5. doi: 10.1006/viro.2002.1358. Virology. 2002. PMID: 12033783
-
Duck and human pandemic influenza A viruses retain sialidase activity under low pH conditions.J Biochem. 2001 Aug;130(2):279-83. doi: 10.1093/oxfordjournals.jbchem.a002983. J Biochem. 2001. PMID: 11481046
-
What adaptive changes in hemagglutinin and neuraminidase are necessary for emergence of pandemic influenza virus from its avian precursor?Biochemistry (Mosc). 2015 Jul;80(7):872-80. doi: 10.1134/S000629791507007X. Biochemistry (Mosc). 2015. PMID: 26542001 Review.
-
Low-pH Stability of Influenza A Virus Sialidase Contributing to Virus Replication and Pandemic.Biol Pharm Bull. 2015;38(6):817-26. doi: 10.1248/bpb.b15-00120. Biol Pharm Bull. 2015. PMID: 26027822 Review.
Cited by
-
Comparison of SARS-CoV-2 spike RNA sequences in feces and nasopharynx indicates intestinal replication.Gut Pathog. 2022 Aug 20;14(1):35. doi: 10.1186/s13099-022-00509-w. Gut Pathog. 2022. PMID: 35987708 Free PMC article.
-
Mimicking the passage of avian influenza viruses through the gastrointestinal tract of chickens.Vet Microbiol. 2019 Dec;239:108462. doi: 10.1016/j.vetmic.2019.108462. Epub 2019 Oct 18. Vet Microbiol. 2019. PMID: 31767100 Free PMC article.
References
-
- Fouchier RA, Munster V, Wallensten A, Bestebroer TM, Herfst S, Smith D, Rimmelzwaan GF, Olsen B, Osterhaus AD. 2005. Characterization of a novel influenza A virus hemagglutinin subtype (H16) obtained from black-headed gulls. J Virol 79:2814–2822. doi:10.1128/JVI.79.5.2814-2822.2005. - DOI - PMC - PubMed
-
- Tong S, Zhu X, Li Y, Shi M, Zhang J, Bourgeois M, Yang H, Chen X, Recuenco S, Gomez J, Chen LM, Johnson A, Tao Y, Dreyfus C, Yu W, McBride R, Carney PJ, Gilbert AT, Chang J, Guo Z, Davis CT, Paulson JC, Stevens J, Rupprecht CE, Holmes EC, Wilson IA, Donis RO. 2013. New World bats harbor diverse influenza A viruses. PLoS Pathog 9:e1003657. doi:10.1371/journal.ppat.1003657. - DOI - PMC - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

