Corticosteroid implants for chronic non-infectious uveitis
- PMID: 26866343
- PMCID: PMC5038923
- DOI: 10.1002/14651858.CD010469.pub2
Corticosteroid implants for chronic non-infectious uveitis
Update in
-
Corticosteroid implants for chronic non-infectious uveitis.Cochrane Database Syst Rev. 2023 Jan 16;1(1):CD010469. doi: 10.1002/14651858.CD010469.pub3. Cochrane Database Syst Rev. 2023. Update in: Cochrane Database Syst Rev. 2023 Aug 29;8:CD010469. doi: 10.1002/14651858.CD010469.pub4. PMID: 36645716 Free PMC article. Updated.
Abstract
Background: Uveitis is a term used to describe a heterogeneous group of intraocular inflammatory diseases of the anterior, intermediate, and posterior uveal tract (iris, ciliary body, choroid). Uveitis is the fifth most common cause of vision loss in high-income countries, accounting for 5% to 20% of legal blindness, with the highest incidence of disease in the working-age population.Corticosteroids are the mainstay of acute treatment for all anatomical subtypes of non-infectious uveitis and can be administered orally, topically with drops or ointments, by periocular (around the eye) or intravitreal (inside the eye) injection, or by surgical implantation.
Objectives: To determine the efficacy and safety of steroid implants in people with chronic non-infectious posterior uveitis, intermediate uveitis, and panuveitis.
Search methods: We searched CENTRAL (which contains the Cochrane Eyes and Vision Trials Register) (Issue 10, 2015), Ovid MEDLINE, Ovid MEDLINE In-Process and Other Non-Indexed Citations, Ovid MEDLINE Daily, Ovid OLDMEDLINE (January 1946 to November 2015), EMBASE (January 1980 to November 2015), PubMed (1948 to November 2015), Latin American and Caribbean Health Sciences Literature Database (LILACS) (1982 to November 2015), the metaRegister of Controlled Trials (mRCT) (www.controlled-trials.com) (last searched 15 April 2013), ClinicalTrials.gov (www.clinicaltrials.gov), and the World Health Organization (WHO) International Clinical Trials Registry Platform (ICTRP) (www.who.int/ictrp/search/en). We did not use any date or language restrictions in the electronic search for studies. We last searched the electronic databases on 6 November 2015.We also searched reference lists of included study reports, citation databases, and abstracts and clinical study presentations from professional meetings.
Selection criteria: We included randomized controlled trials comparing either fluocinolone acetonide (FA) or dexamethasone intravitreal implants with standard-of-care therapy with at least six months of follow-up after treatment. We included studies that enrolled participants of all ages who had chronic non-infectious posterior uveitis, intermediate uveitis, or panuveitis with vision that was better than hand-motion.
Data collection and analysis: Two review authors independently reviewed studies for inclusion. Two review authors independently extracted data and assessed the risk of bias for each study.
Main results: We included data from two studies (619 eyes of 401 participants) that compared FA implants with standard-of-care therapy. Both studies used similar standard-of-care therapy that included administration of prednisolone and, if needed, immunosuppressive agents. The studies included participants from Australia, France, Germany, Israel, Italy, Portugal, Saudi Arabia, Spain, Switzerland, Turkey, the United Kingdom, and the United States. We assessed both studies at high risk of performance and detection bias.Only one study reported our primary outcome, recurrence of uveitis at any point during the study through 24 months. The evidence, judged as moderate-quality, showed that a FA implant probably prevents recurrence of uveitis compared with standard-of-care therapy (risk ratio (RR) 0.29, 95% confidence interval (CI) 0.14 to 0.59; 132 eyes). Both studies reported safety outcomes, and moderate-quality evidence showed increased risks of needing cataract surgery (RR 2.98, 95% CI 2.33 to 3.79; 371 eyes) and surgery to lower intraocular pressure (RR 7.48, 95% CI 3.94 to 14.19; 599 eyes) in the implant group compared with standard-of-care therapy through two years of follow-up. No studies compared dexamethasone implants with standard-of-care therapy.
Authors' conclusions: After considering both benefits and harms reported from two studies in which corticosteroids implants were compared with standard-of-care therapy, we are unable to conclude that the implants are superior to traditional systemic therapy for the treatment of non-infectious uveitis. These studies exhibited heterogeneity in design and outcomes that measured efficacy. Pooled findings regarding safety outcomes suggest increased risks of post-implant surgery for cataract and high intraocular pressure compared with standard-of-care therapy.
Conflict of interest statement
CJB: none known ACV: none known HAL: none known ER: none known RR: consultancy and speakership for Allergan on projects with no relevance to current study PCS: none known SJG: principal investigator of the Allergan Ozurdex Uveitis Trial at Wills Eye Institute JT: none known
Figures
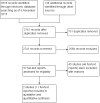










References
References to studies included in this review
Kempen 2011 {published data only}
-
- Kempen JH, Altaweel MM, Holbrook JT, Jabs DA, Louis TA, Sugar EA, et al. Randomized comparison of systemic anti‐inflammatory therapy versus fluocinolone acetonide implant for intermediate, posterior, and panuveitis: the multicenter uveitis steroid treatment trial. Ophthalmology 2011;118(10):1916‐26. - PMC - PubMed
Pavesio 2010 {published data only}
-
- Pavesio C, Zierhut M, Bairi K, Comstock TL, Usner DW. Evaluation of an intravitreal fluocinolone acetonide implant versus standard systemic therapy in noninfectious posterior uveitis. Ophthalmology 2010; Vol. 117, issue 3:567‐75. - PubMed
-
- Pavesio CE, Bairi K. Evaluation of an intravitreal fluocinolone acetonide implant vs. standard systemic therapy in noninfectious posterior uveitis. American Academy of Ophthalmology. 2006:194. - PubMed
References to studies excluded from this review
Acharya 2004 {published data only}
-
- Acharya N, Young L. Sustained‐release drug implants for the treatment of intraocular disease. International Ophthalmology Clinics 2004;44(3):33‐9. - PubMed
Anonymous 1995 {published data only}
-
- Anonymous. Good results from eye implant study. Treatment Review 1995, (16):4. - PubMed
Ansari 2010 {published data only}
Arcinue 2013 {published data only}
-
- Arcinue CA, Ceron OM, Foster CS. A comparison between the fluocinolone acetonide (Retisert) and dexamethasone (Ozurdex) intravitreal implants in uveitis. Journal of Ocular Pharmacology and Therapeutics 2013;29(5):501‐7. - PubMed
Bollinger 2009 {published data only}
-
- Bollinger KE, Smith SD. Prevalence and management of elevated intraocular pressure after placement of an intravitreal sustained‐release steroid implant. Current Opinion in Ophthalmology 2009;20(2):99‐103. - PubMed
Callanan 2008a {published data only}
-
- Callanan DG, Jaffe GJ, Martin DF, Pearson PA, Comstock TL. Treatment of posterior uveitis with a fluocinolone acetonide implant: three‐year clinical trial results. Archives of Ophthalmology 2008;126(9):1191‐201. - PubMed
Campochiaro 2013 {published data only}
-
- Campochiaro PA, Nguyen QD, Hafiz G, Bloom S, Brown DM, Busquets M, et al. Aqueous levels of fluocinolone acetonide after administration of fluocinolone acetonide inserts or fluocinolone acetonide implants. Ophthalmology 2013;120(3):583‐7. - PubMed
Cano‐Parra 2006 {published data only}
-
- Cano‐Parra J, Diaz‐Llopis M. New drugs in the treatment of noninfectious uveitis. Archivos de la Sociedad Espanola de Oftalmologia 2006;81(12):671‐3. - PubMed
CTRI/2014/07/004726 {unpublished data only}
-
- CTRI/2014/12/004726. To assess the safety & efficacy of fluocinolone acetonide intravitreal (FAI) insert in patients with chronic, non‐infectious uveitis affecting the posterior segment of the eye. www.ctri.nic.in/Clinicaltrials/pmaindet2.php?trialid=8285 accessed 15 January 2016.
CTRI/2014/12/005337 {unpublished data only}
-
- CTRI/2014/07/005337. A research to study the effect of fluocinolone Aaetonide intravitreal (FAI) insert in patients with inflammation of middle layer of the eye. http://www.ctri.nic.in/Clinicaltrials/pmaindet2.php?trialid=9684 (accessed 15 January 2016).
Eng 2007 {published data only}
-
- Eng K, Kertes P. Randomized controlled study of an intravitreous dexamethasone drug delivery system in patients with persistent macular edema: Commentary. Evidence‐Based Ophthalmology 2007;8(3):170‐2. - PubMed
Ermakova 2003 {published data only}
-
- Ermakova NA. Efficacy of corticosteroids and cyclosporin in the treatment of retinal vasculitis in patients with Behcet's disease. Advances in Experimental Medicine and Biology 2003;528:563‐5. - PubMed
Galor 2007 {published data only}
-
- Galor A, Margolis R, Kaiser PK, Lowder CY. Vitreous band formation and the sustained‐release, intravitreal fluocinolone (Retisert) implant. Archives of Ophthalmology 2007;125(6):836‐8. - PubMed
Garg 2006 {published data only}
-
- Garg S. Fluocinolone acetonide implant (Retisert) for noninfectious posterior uveitis: Thirty‐four‐week results of a multicenter randomized clinical study. Evidence‐Based Ophthalmology 2006;7(4):216‐7. - PubMed
Goldstein 2007 {published data only}
-
- Goldstein DA, Godfrey DG, Hall A, Callanan DG, Jaffe GJ, Pearson PA, et al. Intraocular pressure in patients with uveitis treated with fluocinolone acetonide implants. Archives of Ophthalmology 2007; Vol. 125, issue 11:1478‐85. - PubMed
Ibrahim 2009 {published data only}
-
- Ibrahim M, Nguyen QD. Treatment of posterior uveitis with a fluocinolone acetonide implant. Evidence‐Based Ophthalmology 2009;10(4):224‐6.
Jaffe 2000a {published data only}
-
- Jaffe GJ, Ben‐Nun J, Guo H, Dunn JP, Ashton P. Fluocinolone acetonide sustained drug delivery device to treat severe uveitis. Ophthalmology 2000; Vol. 107, issue 11:2024‐33. - PubMed
Jaffe 2000b {published data only}
-
- Jaffe GJ, Pearson PA, Ashton P. Dexamethasone sustained drug delivery implant for the treatment of severe uveitis. Retina 2000;20:402‐3. - PubMed
Jaffe 2005a {published data only}
-
- Jaffe GJ, Martin D, Callanan D, Levy B. Fluocinolone acetonide intravitreal implant to treat posterior uveitis: 2‐year results of a multi‐center clinical trial. Investigative Ophthalmology and Visual Science 2005;46(13):ARVO E‐abstract 2386.
Jaffe 2005b {published data only}
-
- Jaffe GJ, McCallum RM, Branchaud B, Skalak C, Butuner Z, Ashton P. Long‐term follow‐up results of a pilot trial of a fluocinolone acetonide implant to treat posterior uveitis. Ophthalmology 2005; Vol. 112, issue 7:1192‐8. - PubMed
Jaffe 2006a {published data only}
-
- Jaffe GJ, Martin D, Callanan D, Pearson PA, Levy B, Comstock T. Fluocinolone acetonide implant (Retisert) for noninfectious posterior uveitis: thirty‐four‐week results of a multicenter randomized clinical study. Ophthalmology 2006; Vol. 113, issue 6:1020‐7. - PubMed
Jaffe 2006b {published data only}
-
- Jaffe GJ, Martin DF, Callanan D, Levy B, Comstock T. Fluocinolone acetonide intravitreal implant to treat posterior segment uveitis: 3‐year results of a multi‐center clinical trial. Investigative Ophthalmology and Visual Science 2006;47(13):ARVO E‐abstract 1523.
Kim 2011 {published data only}
-
- Kim SJ. Diagnosis and management of noninfectious pediatric uveitis. International Ophthalmology Clinics 2011;51(1):129‐45. - PubMed
Kuppermann 2007 {published data only}
-
- Kuppermann BD, Blumenkranz MS, Haller JA, Williams GA, Weinberg DV, Chou C, et al. Randomized controlled study of an intravitreous dexamethasone drug delivery system in patients with persistent macular edema. Archives of Ophthalmology 2007; Vol. 125, issue 3:309‐17. - PubMed
Lowder 2011a {published data only}
-
- Lightman S, Belfort Jr R, Naik RK, Lowder C, Foster CS, Rentz AM, et al. Vision‐related functioning outcomes of dexamethasone intravitreal implant in noninfectious intermediate or posterior uveitis. Investigative Ophthalmology and Visual Science 2013;54(7):4864‐70. - PubMed
-
- Lowder C, Belfort Jr R, Lightman S, Foster CS, Robinson MR, Schiffman RM, et al. Dexamethasone intravitreal implant for noninfectious intermediate or posterior uveitis. Archives of Ophthalmology 2011;129(5):545‐53. - PubMed
Mercante 2007 {published data only}
-
- Mercante AR, Huang SS, Reddy R. Cystoid macular edema in non‐infectious uveitis treated with fluocinolone acetonide intravitreal implant: 3‐year results of a multi‐center clinical trial. Investigative Ophthalmology and Visual Science 2007;48(13):ARVO E‐Abstract 280.
Muller 2004 {published data only}
-
- Muller B, Orlic N, Pleyer U. Intravitreal fluocinolone‐implant (Retisert) in posterior non‐infectious uveitis — intermediate results after 34 weeks. Klinische Monatsblatter fur Augenheilkunde 2004; Vol. 221, issue Suppl 7:S8.
Mustakallio 1973 {published data only}
-
- Mustakallio A, Kaufman HE, Johnston G, Wilson RS, Roberts MD, Harter JC. Corticosteroid efficacy in postoperative uveitis. Annals of Ophthalmology 1973;5(6):719‐30. - PubMed
Naik 2013 {published data only}
-
- Naik RK, Rentz AM, Foster CS, Lightman S, Belfort R, Lowder C, et al. Normative comparison of patient‐reported outcomes in patients with noninfectious uveitis. JAMA Ophthalmology 2013;131(2):219‐25. - PubMed
NCT01694186 {unpublished data only}
-
- NCT01694186. Safety and efficacy of an injectable fluocinolone acetonide intravitreal insert. clinicaltrials.gov/show/NCT01694186 (accessed 15 January 2016).
NCT02309385 {unpublished data only}
-
- NCT02309385. Safety and efficacy study of DSP‐visulex for the treatment of anterior uveitis. clinicaltrials.gov/show/NCT02309385 (accessed 15 January 2016).
NCT02482129 {unpublished data only}
-
- NCT02482129. Proof of concept study to evaluate safety and efficacy of LME636 in the treatment of acute anterior uveitis. clinicaltrials.gov/show/NCT02482129 (accessed 15 January 2016).
NCT02517619 {unpublished data only}
-
- NCT02517619. Safety and efficacy of iontophoretic dexamethasone phosphate ophthalmic solution in non‐infectious anterior uveitis (EGP‐437‐006). clinicaltrials.gov/show/NCT02517619 (accessed 15 January 2016).
Neger 1996 {published data only}
-
- Neger RE. The eyes have it, too. Positively Aware: the Monthly Journal of the Test Positive Aware Network 1996;7(3):10‐1. - PubMed
Novack 2008 {published data only}
-
- Novack GD. Clinical indications for ophthalmic corticosteroids. Ocular Surface 2008;6(4):199‐202. - PubMed
Ram 2013 {published data only}
-
- Ram J, Gupta A, Gupta V. Intraoperative dexamethasone implant in uveitis patients with cataract undergoing phacoemulsification. Ocular Immunology and Inflammation 2013;21(6):462‐7. - PubMed
Sangwan 2007 {published data only}
-
- Sangwan V S, Paul H A, Usner D W. Safety and efficacy of an intravitreal fluocinolone acetonide implant in subjects with noninfectious uveitis. American Academy of Ophthalmology. 2007:199.
Saraiya 2011 {published data only}
-
- Saraiya NV, Patel SS, Goldstein DA. A report of high intraocular pressure with the dexamethasone intravitreal implant. Archives of Ophthalmology 2011;129(12):1638‐9. - PubMed
Sheppard 2012 {published data only}
Taylor 2012 {published data only}
-
- Taylor SR, Tomkins‐Netzer O, Joshi L, Morarji J, McLoone E, Lightman S. Dexamethasone implant in pediatric uveitis. Ophthalmology 2012;119(11):2412. - PubMed
Viola 2009 {published data only}
-
- Viola F, Staurenghi G, Ratiglia R. Uveitis treated with fluocinolone acetonide implants. Archives of Ophthalmology 2009;127(1):115‐6. - PubMed
Wen 1991 {published data only}
-
- Wen S, Gao R, Hu Z. Comparison of Chinese traditional therapy combined with Western medicine and Western medicine alone in the treatment of uveitis. Eye Science 1991;7(4):205‐8. - PubMed
Williams 2004 {published data only}
-
- Williams G A, Kuppermann B D, Blumenkranz M S, Haller J A, Whitcup S M, Weinberg D V. Treatment of inflammatory macular edema with a dexamethasone posterior‐segment drug delivery system. American Academy of Ophthalmology. 2004:87. - PubMed
Williams 2009 {published data only}
-
- Williams GA, Haller JA, Kuppermann BD, Blumenkranz MS, Weinberg DV, Chou C, et al. Dexamethasone posterior‐segment drug delivery system in the treatment of macular edema resulting from uveitis or Irvine‐Gass syndrome. American Journal of Ophthalmology 2009; Vol. 147, issue 6:1048‐54. - PubMed
Yeh 2008 {published data only}
-
- Yeh S, Nussenblatt RB. Fluocinolone acetonide for the treatment of uveitis: weighing the balance between local and systemic immunosuppression. Archives of Ophthalmology 2008;126(9):1287‐9. - PubMed
Additional references
Bollinger 2011
-
- Bollinger K, Kim J, Lowder CY, Kaiser PK, Smith SD. Intraocular pressure outcome of patients with fluocinolone acetonide intravitreal implant for noninfectious uveitis. Ophthalmology 2011;118(10):1927‐31. - PubMed
Callanan 2008b
-
- Callanan DG, Jaffe GJ, Martin DF, Pearson PA, Comstock TL. Treatment of posterior uveitis with a fluocinolone acetonide implant: three‐year clinical trial results. Archives of Ophthalmology 2008;126(9):1191‐201. - PubMed
Campochiaro 2010
-
- Campochiaro PA, Hafiz G, Shah SM, Bloom S, Brown DM, Busquets M, et al. Sustained ocular delivery of fluocinolone acetonide by an intravitreal insert. Ophthalmology 2010;117(7):1393‐9. - PubMed
Deeks 2011
-
- Deeks JJ, Higgins JPT, Altman DG (editors). Chapter 9: Analysing data and undertaking meta‐analyses. In: Higgins JPT, Green S (editors). Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0 (updated March 2011). The Cochrane Collaboration, 2011. Available from www.cochrane‐handbook.org.
Durrani 2004
-
- Durrani OM, Meads CA, Murray PI. Uveitis: a potentially blinding disease. Ophthalmologica 2004;218(4):223‐36. - PubMed
Glanville 2006
Goldstein 2007a
-
- Goldstein DA, Godfrey DG, Hall A, Callanan DG, Jaffe GJ, Pearson PA, et al. Intraocular pressure in patients with uveitis treated with fluocinolone acetonide implants. Archives of Ophthalmology 2007;125(11):1478‐85. - PubMed
Gritz 2004
-
- Gritz DC, Wong IG. Incidence and prevalence of uveitis in Northern California; the Northern California Epidemiology of Uveitis Study. Ophthalmology 2004;111(3):491‐500. - PubMed
Haller 2010
-
- Haller JA, Bandello F, Belfort Jr R, Blumenkranz MS, Gillies M, Heier J, et al. Randomized, sham‐controlled trial of dexamethasone intravitreal implant in patients with macular edema due to retinal vein occlusion. Ophthalmology 2010;117(6):1134‐46. - PubMed
Haupert 2000
-
- Haupert CL, Jaffe GJ. New and emerging treatments for patients with uveitis. International Ophthalmology Clinics 2000;40(2):205‐20. - PubMed
Higgins 2011
-
- Higgins JPT, Altman DG, Sterne JAC (editors). Chapter 8: Assessing risk of bias in included studies. In: Higgins JPT, Green S (editors). Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0 (updated March 2011). The Cochrane Collaboration, 2011. Available from: www.cochrane‐handbook.org.
Jager 2004
-
- Jager RD, Aiello LP, Patel SC, Cunningham Jr ET. Risks of intravitreous injection: a comprehensive review. Retina 2004;24(5):676‐98. - PubMed
Kempen 2011a
-
- Multicenter Uveitis Steroid Treatment (MUST) Trial Research Group, Kempen JH, Altaweel MM, Holbrook JT, Jabs DA, Louis TA, et al. Randomized comparison of systemic anti‐inflammatory therapy versus fluocinolone acetonide implant for intermediate, posterior, and panuveitis: the Multicenter Uveitis Steroid Treatment trial. Ophthalmology 2011;118(10):1916‐26. - PMC - PubMed
Lowder 2011b
-
- Lowder C, Belfort Jr R, Lightman S, Foster CS, Robinson MR, Schiffman RM, et al. Dexamethasone intravitreal implant for noninfectious intermediate or posterior uveitis. Archives of Ophthalmology 2011;129(5):545‐53. - PubMed
Mohammad 2007
-
- Mohammad DA, Sweet BV, Elner SG. Retisert: is the new advance in treatment of uveitis a good one?. Annals of Pharmacotherapy 2007;41(3):449‐54. - PubMed
Nussenblatt 1990
-
- Nussenblatt RB. The natural history of uveitis. International Ophthalmology 1990;14(5‐6):303‐8. - PubMed
Pavesio 2010a
-
- Pavesio C, Zierhut M, Bairi K, Comstock TL, Usner DW, Fluocinolone Acetonide Study Group. Evaluation of an intravitreal fluocinolone acetonide implant versus standard systemic therapy in noninfectious posterior uveitis. Ophthalmology 2010;117(3):567‐75. - PubMed
RevMan 2014 [Computer program]
-
- The Nordic Cochrane Centre, The Cochrane Collaboration. Review Manager (RevMan). Version 5.3. Copenhagen: The Nordic Cochrane Centre, The Cochrane Collaboration, 2014.
Schünemann 2011
-
- Schünemann HJ, Oxman AD, Vist GE, Higgins JPT, Deeks JJ, et al. Chapter 12: Interpreting results and drawing conclusions. In: Higgins JPT, Green S (editors). Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0 (updated March 2011). The Cochrane Collaboration, 2011. Available from: www.cochrane‐handbook.org.
Suttorp‐Schulten 1996
Taylor 2010
-
- Taylor SR, Isa H, Joshi L, Lightman S. New developments in corticosteroid therapy for uveitis. Ophthalmologica 2010;224(Suppl 1):46‐53. - PubMed
Williams 2009a
-
- Williams GA, Haller JA, Kuppermann BD, Blumenkranz MS, Weinberg DV, Chou C, et al. Dexamethasone posterior‐segment drug delivery system in the treatment of macular edema resulting from uveitis or Irvine‐Gass syndrome. American Journal of Ophthalmology 2009;147(6):1048‐54. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

