Wnt/β-Catenin Signaling Determines the Vasculogenic Fate of Postnatal Mesenchymal Stem Cells
- PMID: 26866635
- PMCID: PMC5338744
- DOI: 10.1002/stem.2334
Wnt/β-Catenin Signaling Determines the Vasculogenic Fate of Postnatal Mesenchymal Stem Cells
Abstract
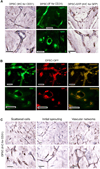
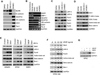
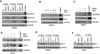
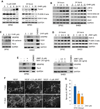
Vasculogenesis is the process of de novo blood vessel formation observed primarily during embryonic development. Emerging evidence suggest that postnatal mesenchymal stem cells are capable of recapitulating vasculogenesis when these cells are engaged in tissue regeneration. However, the mechanisms underlining the vasculogenic differentiation of mesenchymal stem cells remain unclear. Here, we used stem cells from human permanent teeth (dental pulp stem cells [DPSC]) or deciduous teeth (stem cells from human exfoliated deciduous teeth [SHED]) as models of postnatal primary human mesenchymal stem cells to understand mechanisms regulating their vasculogenic fate. GFP-tagged mesenchymal stem cells seeded in human tooth slice/scaffolds and transplanted into immunodeficient mice differentiate into human blood vessels that anastomize with the mouse vasculature. In vitro, vascular endothelial growth factor (VEGF) induced the vasculogenic differentiation of DPSC and SHED via potent activation of Wnt/β-catenin signaling. Further, activation of Wnt signaling is sufficient to induce the vasculogenic differentiation of postnatal mesenchymal stem cells, while Wnt inhibition blocked this process. Notably, β-catenin-silenced DPSC no longer differentiate into endothelial cells in vitro, and showed impaired vasculogenesis in vivo. Collectively, these data demonstrate that VEGF signaling through the canonical Wnt/β-catenin pathway defines the vasculogenic fate of postnatal mesenchymal stem cells. Stem Cells 2016;34:1576-1587.
Keywords: Angiogenesis; Dental pulp stem cells; Multipotency; Self-renewal; Tissue engineering; Vasculogenesis.
© 2016 AlphaMed Press.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

