Synergistic Malaria Parasite Killing by Two Types of Plasmodial Surface Anion Channel Inhibitors
- PMID: 26866812
- PMCID: PMC4750852
- DOI: 10.1371/journal.pone.0149214
Synergistic Malaria Parasite Killing by Two Types of Plasmodial Surface Anion Channel Inhibitors
Abstract
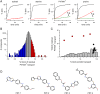
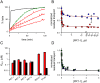
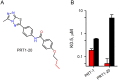
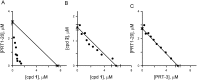
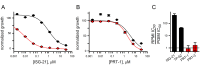
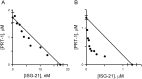
Malaria parasites increase their host erythrocyte's permeability to a broad range of ions and organic solutes. The plasmodial surface anion channel (PSAC) mediates this uptake and is an established drug target. Development of therapies targeting this channel is limited by several problems including interactions between known inhibitors and permeating solutes that lead to incomplete channel block. Here, we designed and executed a high-throughput screen to identify a novel class of PSAC inhibitors that overcome this solute-inhibitor interaction. These new inhibitors differ from existing blockers and have distinct effects on channel-mediated transport, supporting a model of two separate routes for solute permeation though PSAC. Combinations of inhibitors specific for the two routes had strong synergistic action against in vitro parasite propagation, whereas combinations acting on a single route produced only additive effects. The magnitude of synergism depended on external nutrient concentrations, consistent with an essential role of the channel in parasite nutrient acquisition. The identified inhibitors will enable a better understanding of the channel's structure-function and may be starting points for novel combination therapies that produce synergistic parasite killing.
Conflict of interest statement
Figures
Similar articles
-
Solute restriction reveals an essential role for clag3-associated channels in malaria parasite nutrient acquisition.Mol Pharmacol. 2012 Dec;82(6):1104-14. doi: 10.1124/mol.112.081224. Epub 2012 Sep 4. Mol Pharmacol. 2012. PMID: 22949525 Free PMC article.
-
The plasmodial surface anion channel is functionally conserved in divergent malaria parasites.Eukaryot Cell. 2005 Dec;4(12):2153-9. doi: 10.1128/EC.4.12.2153-2159.2005. Eukaryot Cell. 2005. PMID: 16339732 Free PMC article.
-
Changes in the plasmodial surface anion channel reduce leupeptin uptake and can confer drug resistance in Plasmodium falciparum-infected erythrocytes.Antimicrob Agents Chemother. 2008 Jul;52(7):2346-54. doi: 10.1128/AAC.00057-08. Epub 2008 Apr 28. Antimicrob Agents Chemother. 2008. PMID: 18443109 Free PMC article.
-
Why do malaria parasites increase host erythrocyte permeability?Trends Parasitol. 2014 Mar;30(3):151-9. doi: 10.1016/j.pt.2014.01.003. Epub 2014 Feb 5. Trends Parasitol. 2014. PMID: 24507014 Free PMC article. Review.
-
Epigenetics of malaria parasite nutrient uptake, but why?Trends Parasitol. 2022 Aug;38(8):618-628. doi: 10.1016/j.pt.2022.05.005. Epub 2022 May 28. Trends Parasitol. 2022. PMID: 35641406 Free PMC article. Review.
Cited by
-
Plasmodium vivax infection compromises reticulocyte stability.Nat Commun. 2021 Mar 12;12(1):1629. doi: 10.1038/s41467-021-21886-x. Nat Commun. 2021. PMID: 33712609 Free PMC article.
-
Complex nutrient channel phenotypes despite Mendelian inheritance in a Plasmodium falciparum genetic cross.PLoS Pathog. 2020 Feb 18;16(2):e1008363. doi: 10.1371/journal.ppat.1008363. eCollection 2020 Feb. PLoS Pathog. 2020. PMID: 32069335 Free PMC article.
-
Transporter-Mediated Solutes Uptake as Drug Target in Plasmodium falciparum.Front Pharmacol. 2022 Feb 7;13:845841. doi: 10.3389/fphar.2022.845841. eCollection 2022. Front Pharmacol. 2022. PMID: 35370717 Free PMC article. Review.
-
The critical role of PSAC channel in malaria parasite survival is driven home by phenotypic screening under relevant nutrient levels.Cell Chem Biol. 2025 Jun 19;32(6):826-838.e13. doi: 10.1016/j.chembiol.2025.05.001. Epub 2025 May 23. Cell Chem Biol. 2025. PMID: 40412380
-
Targeting Channels and Transporters in Protozoan Parasite Infections.Front Chem. 2018 Mar 27;6:88. doi: 10.3389/fchem.2018.00088. eCollection 2018. Front Chem. 2018. PMID: 29637069 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

