Mesenchymal Stem Cell Alterations in Bone Marrow Lesions in Patients With Hip Osteoarthritis
- PMID: 26866940
- PMCID: PMC4941540
- DOI: 10.1002/art.39622
Mesenchymal Stem Cell Alterations in Bone Marrow Lesions in Patients With Hip Osteoarthritis
Abstract
Objective: In patients with osteoarthritis (OA), bone marrow lesions (BMLs) are intimately linked to disease progression. We hypothesized that aberrant multipotential stromal cell (also known as mesenchymal stem cell [MSC]) responses within bone tissue contributes to BML pathophysiology. The aim of this study was to investigate BML and non-BML native subchondral bone MSCs for numeric, topographic, in vitro functional, and gene expression differences.
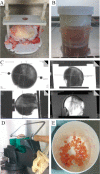
Methods: Ex vivo 3T magnetic resonance imaging (MRI) of the femoral heads of 20 patients with hip OA was performed. MRI-determined BML and non-BML regions were excised and enzymatically treated to extract cells and quantify MSCs using flow cytometry and colony-forming unit-fibroblast (CFU-F) assay. Immunohistochemical analysis was performed to determine in vivo CD271+ MSC distribution. Culture-expanded CD271+ cells were analyzed for tripotentiality and gene expression.
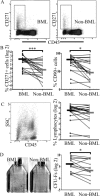
Results: BML regions were associated with greater trabecular bone area and cartilage damage compared with non-BML regions. The proportion of CD45-CD271+ MSCs was higher in BML regions compared with non-BML regions (median difference 5.6-fold; P < 0.001); the CFU-F assay showed a similar trend (median difference 4.3-fold; P = 0.013). Immunohistochemistry revealed CD271+ cell accumulation in bone adjacent to cartilage defects and areas of osteochondral angiogenesis. BML MSCs had lower proliferation and mineralization capacities in vitro and altered expression of TNFSF11/RANKL and CXCR4/stromal cell-derived factor 1 receptor. OA MSCs showed up-regulated transcripts for CXCR1 and CCR6 compared with MSCs derived from healthy or osteoporotic bone.
Conclusion: This study is the first to show numeric and topographic alterations in native MSCs in the diseased bone of patients with hip OA. Given the associated functional perturbation of MSCs, these data suggest that subchondral bone MSC manipulation may be an OA treatment target.
© 2016 The Authors. Arthritis & Rheumatology published by Wiley Periodicals, Inc. on behalf of the American College of Rheumatology.
Figures




References
-
- Nho SJ, Kymes SM, Callaghan JJ, Felson DT. The burden of hip osteoarthritis in the United States: epidemiologic and economic considerations. J Am Acad Orthop Surg 2013;21 Suppl 1:S1–6. - PubMed
-
- Mansell JP, Collins C, Bailey AJ. Bone, not cartilage, should be the major focus in osteoarthritis. Nat Clin Pract Rheumatol 2007;3:306–7. - PubMed
-
- Goldring MB, Goldring SR. Articular cartilage and subchondral bone in the pathogenesis of osteoarthritis. Ann N Y Acad Sci 2010;1192:230–7. - PubMed
-
- Felson DT, Chaisson CE, Hill CL, Totterman SM, Gale ME, Skinner KM, et al. The association of bone marrow lesions with pain in knee osteoarthritis. Ann Intern Med 2001;134:541–9. - PubMed
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

