Beta-arrestin 2 rather than G protein efficacy determines the anxiolytic-versus antidepressant-like effects of nociceptin/orphanin FQ receptor ligands
- PMID: 26867504
- PMCID: PMC5567672
- DOI: 10.1016/j.neuropharm.2016.02.003
Beta-arrestin 2 rather than G protein efficacy determines the anxiolytic-versus antidepressant-like effects of nociceptin/orphanin FQ receptor ligands
Abstract
Background and purpose: Nociceptin/orphanin FQ (N/OFQ) receptor (NOP) agonists produce anxiolytic-like effects in rodents while antagonists promote antidepressant-like effects. The aim of this study was to investigate the effect on anxiety and depression of NOP receptor partial agonists such as the peptides [F/G]N/OFQ(1-13)NH2 and UFP-113 and the non-peptide AT-090.
Experimental approach: In vitro AT-090, UFP-113, and [F/G]N/OFQ(1-13)NH2 were tested for their ability to promote NOP/G-protein and NOP/β-arrestin 2 interaction, using a bioluminescence resonance energy transfer assay. In vivo, they were tested in mice in the elevated plus maze (EPM) and in the forced swim (FST) tests. NOP partial agonists effects were systematically compared to those of full agonists (N/OFQ and Ro 65-6570) and antagonists (UFP-101 and SB-612111).
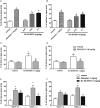
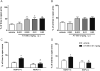
Key results: In vitro, AT-090, UFP-113, and [F/G]N/OFQ(1-13)NH2 promoted NOP/G protein interaction, with maximal effects lower than those evoked by N/OFQ and Ro 65-6570. AT-090 behaved as a NOP partial agonist also in inducing β-arrestin 2 recruitment, while UFP-113 and [F/G]N/OFQ(1-13)NH2 were inactive in this assay. In vivo, AT-090 induced anxiolytic-like effects in the EPM but was inactive in the FST. Opposite results were obtained with UFP-113 and [F/G]N/OFQ(1-13)NH2.
Conclusions and implications: NOP ligands producing similar effects on NOP/G protein interaction (partial agonism) but showing different effects on β-arrestin 2 recruitment (partial agonism vs antagonism) elicited different actions on anxiety and mood. These results suggest that the action of a NOP ligand on emotional states is better predicted based on its β-arrestin 2 rather than G-protein efficacy.
Keywords: Anxiety; BRET; Depression; Elevated plus maze; Forced swim test; G protein; Mouse; N/OFQ; N/OFQ (PubChem CID: 16131448); NOP receptor partial agonist; SB-612111 (PubChem CID: 10047612); UFP-101 (PubChem CID: 25081457); β-arrestin.
Copyright © 2016 Elsevier Ltd. All rights reserved.
Conflict of interest statement
All other authors declare no conflicts of interest.
Figures
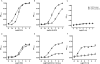
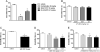
References
-
- Arduin M, Spagnolo B, Calo G, Guerrini R, Carra G, Fischetti C, et al. Synthesis and biological activity of nociceptin/orphanin FQ analogues substituted in position 7 or 11 with Calpha, alpha-dialkylated amino acids. Bioorg Med. Chem. 2007;15(13):4434–4443. - PubMed
-
- Attramadal H, Arriza JL, Aoki C, Dawson TM, Codina J, Kwatra MM, et al. Beta-arrestin2, a novel member of the arrestin/beta-arrestin gene family. J. Biol. Chem. 1992;267(25):17882–17890. - PubMed
-
- Avissar S, Matuzany-Ruban A, Tzukert K, Schreiber G. Beta-arrestin-1 levels: reduced in leukocytes of patients with depression and elevated by antidepressants in rat brain. Am. J. Psychiatry. 2004;161(11):2066–2072. - PubMed
-
- Beaulieu JM, Gainetdinov RR, Caron MG. Akt/GSK3 signaling in the action of psychotropic drugs. Annu. Rev. Pharmacol. Toxicol. 2009;49:327–347. - PubMed
-
- Beaulieu JM, Marion S, Rodriguiz RM, Medvedev IO, Sotnikova TD, Ghisi V, et al. A beta-arrestin 2 signaling complex mediates lithium action on behavior. Cell. 2008;132(1):125–136. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

