Bone marrow-derived mesenchymal stromal cells differ in their attachment to fibronectin-derived peptides from term placenta-derived mesenchymal stromal cells
- PMID: 26869043
- PMCID: PMC4751672
- DOI: 10.1186/s13287-015-0243-6
Bone marrow-derived mesenchymal stromal cells differ in their attachment to fibronectin-derived peptides from term placenta-derived mesenchymal stromal cells
Abstract
Introduction: Human mesenchymal stromal cells (MSCs) can be isolated from different sources including bone marrow and term placenta. These two populations display distinct patterns of proliferation and differentiation in vitro. Since proliferation and differentiation of cells are modulated by cell-matrix interactions, we investigated the attachment of MSCs to a set of peptide-coated surfaces and explored their interactions with peptides in suspension.
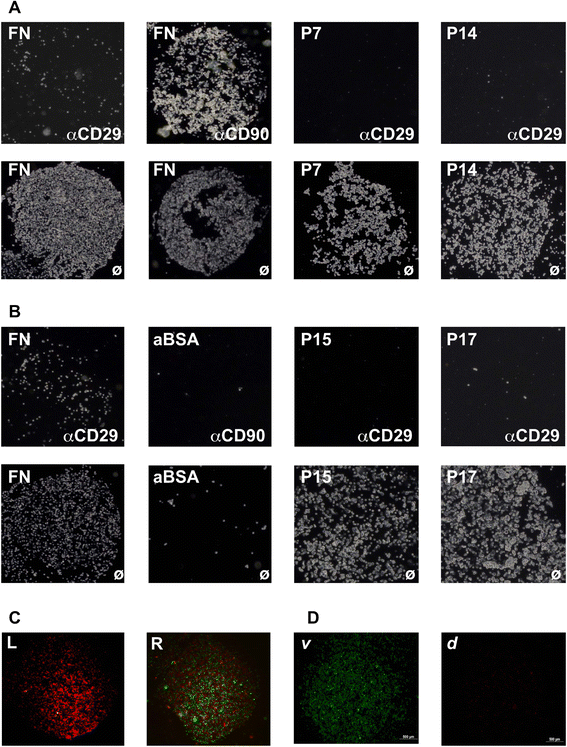
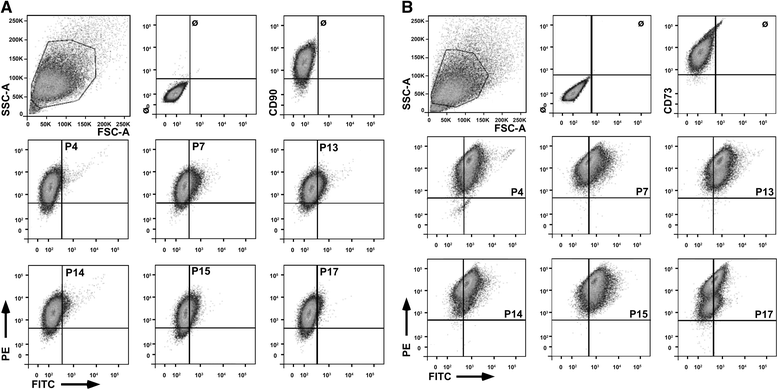
Methods: Human MSCs were isolated from bone marrow and term placenta and expanded. Binding of MSCs to peptides was investigated by a cell-attachment spot assay, by blocking experiments and flow cytometry. The integrin expression pattern was explored by a transcript array and corroborated by quantitative reverse transcription polymerase chain reaction and flow cytometry.
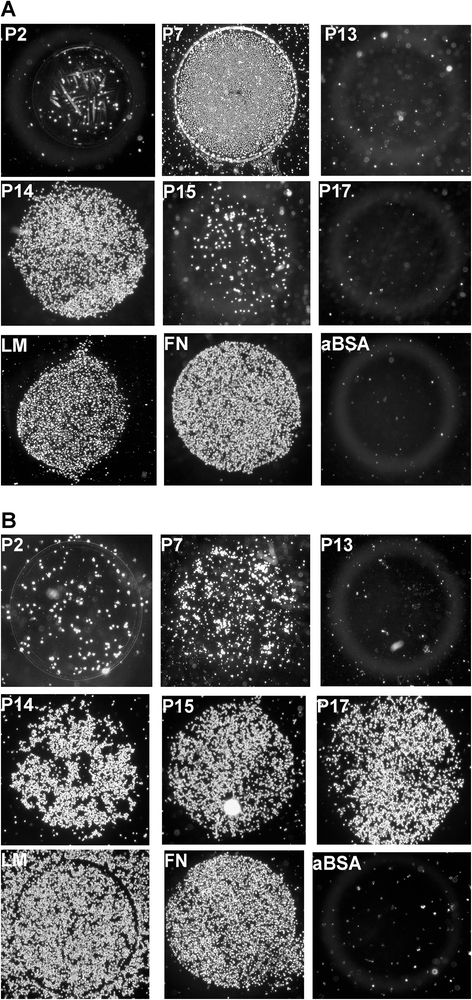
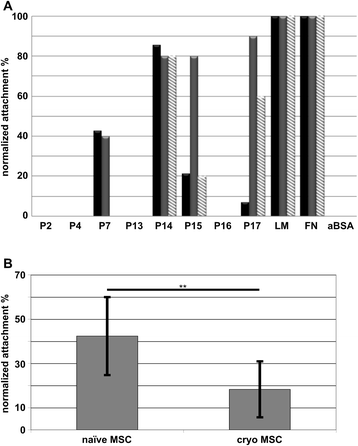
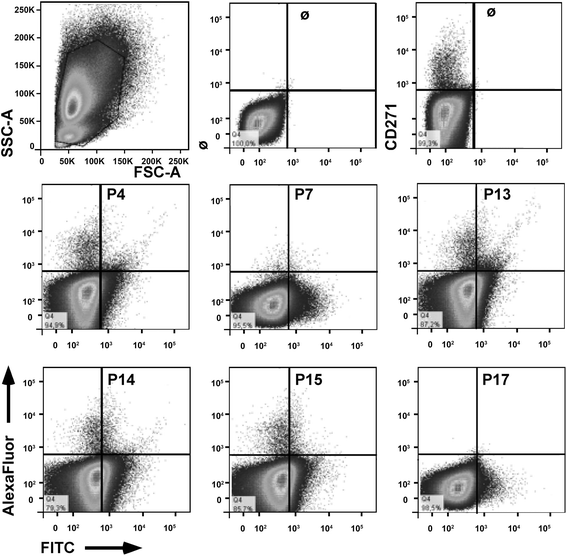
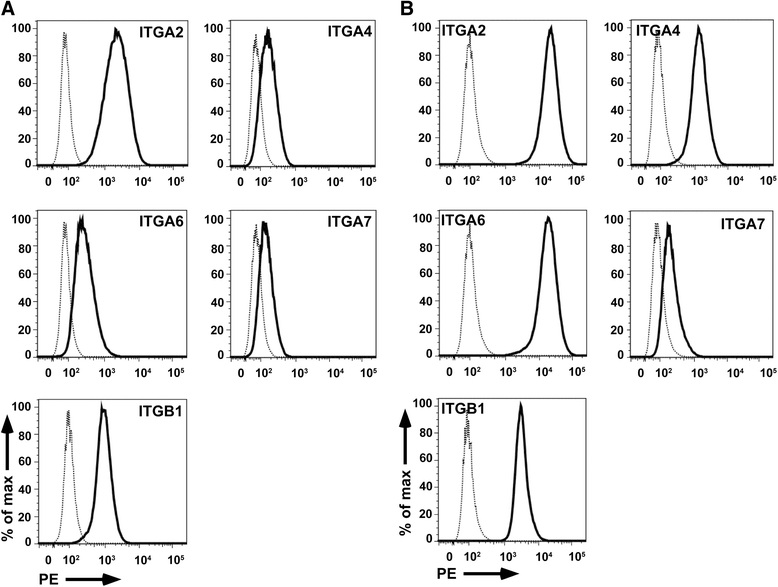
Results: Expanded placenta-derived MSCs (pMSCs) attached well to surfaces coated with fibronectin-derived peptides P7, P15, and P17, whereas bone marrow-derived MSCs (bmMSCs) attached to P7, but barely to P15 and P17. The binding of bmMSCs and pMSCs to the peptides was mediated by β1 integrins. In suspension, expanded bmMSCs barely bind to P7, P13, P15, and less to P14 and P17. Ex vivo, bmMSCs failed to bind P7, but displayed a weak interaction with P13, P14, and P15. In suspension, expanded pMSCs displayed binding to many peptides, including P4, P7, P13, P14, P15, and P17. The differences observed in binding of bmMSCs and pMSCs to the peptides were associated with significant differences in expression of integrin α2-, α4-, and α6-chains.
Conclusions: Human bmMSCs and pMSCs show distinct patterns of attachment to defined peptides and maintain differences in expression of integrins in vitro. Interactions of ex vivo bmMSCs with a given peptide yield different staining patterns compared to expanded bmMSCs in suspension. Attachment of expanded MSCs to peptides on surfaces is different from interactions of expanded MSCs with peptides in suspension. Studies designed to investigate the interactions of human MSCs with peptide-augmented scaffolds or peptides in suspension must therefore regard these differences in cell-peptide interactions.
Figures
Similar articles
-
Allogenic Use of Human Placenta-Derived Stromal Cells as a Highly Active Subtype of Mesenchymal Stromal Cells for Cell-Based Therapies.Int J Mol Sci. 2021 May 18;22(10):5302. doi: 10.3390/ijms22105302. Int J Mol Sci. 2021. PMID: 34069909 Free PMC article. Review.
-
Human Placenta-Derived CD146-Positive Mesenchymal Stromal Cells Display a Distinct Osteogenic Differentiation Potential.Stem Cells Dev. 2015 Jul 1;24(13):1558-69. doi: 10.1089/scd.2014.0465. Epub 2015 Apr 13. Stem Cells Dev. 2015. PMID: 25743703
-
Expression of Desmoglein 2, Desmocollin 3 and Plakophilin 2 in Placenta and Bone Marrow-Derived Mesenchymal Stromal Cells.Stem Cell Rev Rep. 2017 Apr;13(2):258-266. doi: 10.1007/s12015-016-9710-4. Stem Cell Rev Rep. 2017. PMID: 28154962
-
Comparative phenotypic transcriptional characterization of human full-term placenta-derived mesenchymal stromal cells compared to bone marrow-derived mesenchymal stromal cells after differentiation in myogenic medium.Placenta. 2017 Jan;49:64-67. doi: 10.1016/j.placenta.2016.11.007. Epub 2016 Nov 16. Placenta. 2017. PMID: 28012456
-
Immunomodulatory properties of human placental mesenchymal stem/stromal cells.Placenta. 2017 Nov;59:87-95. doi: 10.1016/j.placenta.2017.04.003. Epub 2017 Apr 7. Placenta. 2017. PMID: 28411943 Review.
Cited by
-
Allogenic Use of Human Placenta-Derived Stromal Cells as a Highly Active Subtype of Mesenchymal Stromal Cells for Cell-Based Therapies.Int J Mol Sci. 2021 May 18;22(10):5302. doi: 10.3390/ijms22105302. Int J Mol Sci. 2021. PMID: 34069909 Free PMC article. Review.
-
Wharton's Jelly Mesenchymal Stromal Cells from Human Umbilical Cord: a Close-up on Immunomodulatory Molecules Featured In Situ and In Vitro.Stem Cell Rev Rep. 2019 Dec;15(6):900-918. doi: 10.1007/s12015-019-09907-1. Stem Cell Rev Rep. 2019. PMID: 31741193
-
Pericellular collagen I coating for enhanced homing and chondrogenic differentiation of mesenchymal stem cells in direct intra-articular injection.Stem Cell Res Ther. 2018 Jun 27;9(1):174. doi: 10.1186/s13287-018-0916-z. Stem Cell Res Ther. 2018. PMID: 29945671 Free PMC article.
-
Scalable manufacturing of gene-modified human mesenchymal stromal cells with microcarriers in spinner flasks.Appl Microbiol Biotechnol. 2023 Sep;107(18):5669-5685. doi: 10.1007/s00253-023-12634-w. Epub 2023 Jul 20. Appl Microbiol Biotechnol. 2023. PMID: 37470820 Free PMC article.
-
Global phenotypic characterisation of human platelet lysate expanded MSCs by high-throughput flow cytometry.Sci Rep. 2018 Mar 2;8(1):3907. doi: 10.1038/s41598-018-22326-5. Sci Rep. 2018. PMID: 29500387 Free PMC article.
References
-
- Pittenger MF, Mosca JD, McIntosh KR. Human mesenchymal stem cells: progenitor cells for cartilage, bone, fat and stroma. Curr Top Microbiol Immunol. 2000;251:3–11. - PubMed
-
- Kerkis I, Kerkis A, Dozortsev D, Stukart-Parsons GC, Gomes Massironi SM, Pereira LV, et al. Isolation and characterization of a population of immature dental pulp stem cells expressing OCT-4 and other embryonic stem cell markers. Cells Tissues Organs. 2006;184(3–4):105–16. doi: 10.1159/000099617. - DOI - PubMed
-
- Alexander D, Schäfer F, Munz A, Friedrich B, Klein C, Hoffmann J, et al. NGFR: a new osteogenic differentiation marker in mineralizing periosteal cells. Tissue Eng. 2009;15(3):715.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

