A synergistic blocking effect of Mg²⁺ and spermine on the inward rectifier K⁺ (Kir2.1) channel pore
- PMID: 26869275
- PMCID: PMC4751470
- DOI: 10.1038/srep21493
A synergistic blocking effect of Mg²⁺ and spermine on the inward rectifier K⁺ (Kir2.1) channel pore
Abstract
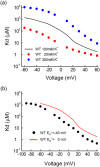
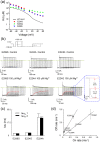
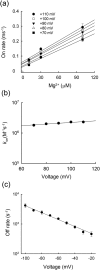
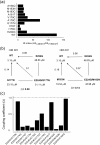
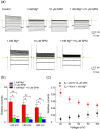
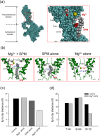
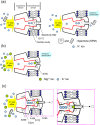
Inward rectifier K(+) channels (Kir2.1) exhibit an extraordinary rectifying feature in the current-voltage relationship. We have previously showed that the bundle-crossing region of the transmembrane domain constitutes the crucial segment responsible for the polyamine block. In this study, we demonstrated that the major blocking effect of intracellular Mg(2+) on Kir2.1 channels is also closely correlated with K(+) current flow, and the coupled movements of Mg(2+) and K(+) seem to happen in the same flux-coupling segment of the pore as polyamines. With a preponderant outward K(+) flow, intracellular Mg(2+) would also be pushed to and thus stay at the outermost site of a flux-coupling segment in the bundle-crossing region of Kir2.1 channels to block the pore, although with a much lower apparent affinity than spermine (SPM). However, in contrast to the evident possibilities of outward exit of SPM through the channel pore especially during strong membrane depolarization, intracellular Mg(2+) does not seem to traverse the Kir2.1 channel pore in any case. Intracellular Mg(2+) and SPM therefore may have a synergistic action on the pore-blocking effect, presumably via prohibition of the outward exit of the higher-affinity blocking SPM by the lower-affinity Mg(2+).
Figures




Similar articles
-
The bundle crossing region is responsible for the inwardly rectifying internal spermine block of the Kir2.1 channel.Pflugers Arch. 2014 Feb;466(2):275-93. doi: 10.1007/s00424-013-1322-0. Epub 2013 Jul 20. Pflugers Arch. 2014. PMID: 23873351
-
Gating of the kir2.1 channel at the bundle crossing region by intracellular spermine and other cations.J Cell Physiol. 2014 Nov;229(11):1703-21. doi: 10.1002/jcp.24616. J Cell Physiol. 2014. PMID: 24633623
-
Two modes of polyamine block regulating the cardiac inward rectifier K+ current IK1 as revealed by a study of the Kir2.1 channel expressed in a human cell line.J Physiol. 2004 Apr 1;556(Pt 1):61-78. doi: 10.1113/jphysiol.2003.055434. Epub 2004 Jan 14. J Physiol. 2004. PMID: 14724206 Free PMC article.
-
Spermine is fit to block inward rectifier (Kir) channels.J Gen Physiol. 2003 Nov;122(5):481-4. doi: 10.1085/jgp.200308957. J Gen Physiol. 2003. PMID: 14581580 Free PMC article. Review. No abstract available.
-
Mechanism of rectification in inward-rectifier K+ channels.Annu Rev Physiol. 2004;66:103-29. doi: 10.1146/annurev.physiol.66.032102.150822. Annu Rev Physiol. 2004. PMID: 14977398 Review.
Cited by
-
The Biophysical Basis Underlying Gating Changes in the p.V1316A Mutant Nav1.7 Channel and the Molecular Pathogenesis of Inherited Erythromelalgia.PLoS Biol. 2016 Sep 21;14(9):e1002561. doi: 10.1371/journal.pbio.1002561. eCollection 2016 Sep. PLoS Biol. 2016. PMID: 27653502 Free PMC article.
-
Anomalous enhancement of resurgent Na+ currents at high temperatures by SCN9A mutations underlies the episodic heat-enhanced pain in inherited erythromelalgia.Sci Rep. 2019 Aug 22;9(1):12251. doi: 10.1038/s41598-019-48672-6. Sci Rep. 2019. PMID: 31439884 Free PMC article.
-
Inwardly rectifying potassium channels: Critical insights for insect species and Apis mellifera.Channels (Austin). 2025 Dec;19(1):2529250. doi: 10.1080/19336950.2025.2529250. Epub 2025 Jul 10. Channels (Austin). 2025. PMID: 40641062 Free PMC article. Review.
-
Excitation and electroporation in genetically engineered excitable S-HEK cells exposed to electric pulses of different durations.Sci Rep. 2025 Jul 2;15(1):23451. doi: 10.1038/s41598-025-06989-5. Sci Rep. 2025. PMID: 40603954 Free PMC article.
-
Genetically engineered HEK cells as a valuable tool for studying electroporation in excitable cells.Sci Rep. 2024 Jan 6;14(1):720. doi: 10.1038/s41598-023-51073-5. Sci Rep. 2024. PMID: 38184741 Free PMC article.
References
-
- Bichet D., Haass F. A. & Jan L. Y. Merging functional studies with structures of inward-rectifier K(+) channels. Nat Rev Neurosci 4, 957–967 (2003). - PubMed
-
- Lu T., Nguyen B., Zhang X. & Yang J. Architecture of a K+ channel inner pore revealed by stoichiometric covalent modification. Neuron 22, 571–580 (1999). - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

