Helicobacter pylori-infected MSCs acquire a pro-inflammatory phenotype and induce human gastric cancer migration by promoting EMT in gastric cancer cells
- PMID: 26870232
- PMCID: PMC4726974
- DOI: 10.3892/ol.2015.3897
Helicobacter pylori-infected MSCs acquire a pro-inflammatory phenotype and induce human gastric cancer migration by promoting EMT in gastric cancer cells
Abstract
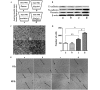
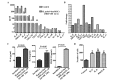
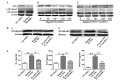
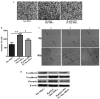
Accumulating clinical and experimental evidence has suggested that Helicobacter pylori (H. pylori) infection-associated gastric cancer (GC) is associated with high rates of mortality and serious health effects. The majority of patients succumb to H. pylori infection-associated GC due to metastasis. Mesenchymal stem cells (MSCs), which have multipotent differentiation potential, may be recruited into the tumor-associated stroma. MSCs are crucial components of the H. pylori infection-associated GC microenvironment, and may be critical for GC cell migration. In this study, an MSCs/H. pylori co-culture model was designed, and the effect of H. pylori-infected MSCs on the migration of GC cells was evaluated using a Transwell migration assay. H. pylori-infected MSC cytokine expression was evaluated using Luminex/ELISA. The expression of epithelial-mesenchymal transition (EMT) markers in the GC cells treated with supernatants from H. pylori-infected MSCs were detected by western blot analysis. The results demonstrated that the interaction between MSCs and H. pylori may induce GC cell migration, through secretion of a combination of cytokines that promote EMT in GC cells. The expression of phosphorylated forms of nuclear factor-κB (NF-κB) was observed to be increased in MSCs by H. pylori. Inhibition of NF-κB activation by pyrrolidine dithiocarbamate blocked the effects of H. pylori-infected MSCs on SGC-7901 human stomach adenocarcinoma cell migration. Overall, the results of the present study suggest that H. pylori-infected MSCs acquire a pro-inflammatory phenotype through secretion of a combination of multiple cytokines, a number of which are NF-κB-dependent. These cytokines enhance H. pylori infection-associated GC cell migration by promoting EMT in GC cells. The results of the present study provide novel evidence for the modulatory effect of MSCs in the tumor microenvironment and provide insight into the significance of stromal cell involvement in GC progression.
Keywords: Helicobacter pylori; epithelial-mesenchymal transition; gastric cancer; mesenchymal stem cells; migration.
Figures
References
-
- Liu GM, Zhou C, Xie C, Yang Z, Lv NH. Recent advances in research of gastric cancer stem cells. World Chinese Journal of Digestology. 2012;7:574–579. (In Chinese)
-
- Lee YY, Derakhshan MH. Environmental and lifestyle risk factors of gastric cancer. Arch Iran Med. 2013;16:358–365. - PubMed
-
- Nitta T, Mitsuhashi T, Hatanaka Y, Miyamoto M, Oba K, Tsuchikawa T, Suzuki Y, Hatanaka KC, Hirano S, Matsuno Y. Prognostic significance of epithelial-mesenchymal transition-related markers in extrahepatic cholangiocarcinoma: Comprehensive immunohistochemical study using a tissue microarray. Br J Cancer. 2014;111:1363–1372. doi: 10.1038/bjc.2014.415. - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous
